CBIC Certified Infection Control Exam Questions and Answers
The MOST common reason for contamination of compounded pharmaceutical products is:
Options:
Direct touch by personnel
Inadequate laminar airflow
Infrequent environmental sampling
Inappropriate storage of pharmaceutical items
Answer:
AExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies direct touch contamination by personnel as the most common cause of contamination of compounded pharmaceutical products. Human contact—particularly hands, gloves, sleeves, or improper manipulation of sterile components—is the greatest source of microbial contamination during compounding activities.
Even when engineering controls such as laminar airflow workbenches and cleanrooms are functioning correctly, contamination can occur if aseptic technique is not strictly followed. Touching sterile vial stoppers, syringe tips, needle hubs, or critical sites with nonsterile hands or gloves introduces microorganisms directly into the product. The Study Guide emphasizes that aseptic technique, hand hygiene, glove use, and competency validation are essential to preventing contamination.
Option B, inadequate laminar airflow, can contribute to contamination but is less common than direct touch errors and is usually detected through certification and monitoring. Option C, infrequent environmental sampling, does not cause contamination but may delay detection of problems. Option D, inappropriate storage, can affect product stability but is not the primary cause of contamination during compounding.
For CIC® exam preparation, it is critical to recognize that human factors are the leading source of contamination in sterile compounding. Infection prevention strategies therefore focus heavily on staff training, competency assessment, observation, and adherence to aseptic technique standards to reduce contamination risk.
An infection preventionist (IP) is asked to evaluate a series of published sources about CAUTI prevention strategies. Which source shows the strongest evidence for the IP to implement change?
Options:
A randomized controlled trial from another hospital
Evidence from reports written by authorities who are on expert committees
Data from a case study from a well-designed case-control or cohort study
A systematic review of relevant controlled studies and evidence-based practices
Answer:
DExplanation:
When an IP is selecting evidence to support practice change, the “strength” of evidence is typically judged using an evidence hierarchy. In most evidence pyramids, systematic reviews (often with meta-analysis) of well-designed studies sit at or near the top because they use explicit methods to search for, appraise, and synthesize findings across multiple studies—reducing the influence of chance results and individual-study bias.
Option D is therefore strongest: a systematic review of relevant controlled studies and evidence-based practices provides the most robust overall summary for decision-making compared with any single study. Randomized controlled trials (option A) are strong primary studies, but they represent one setting/population and can be affected by local factors; a high-quality systematic review places RCTs in context and evaluates consistency across multiple trials.
Observational designs (option C, cohort/case-control) are generally lower in the hierarchy for intervention effectiveness due to confounding risk, and expert committee reports (option B) are typically considered lower-level evidence unless they are explicitly based on systematic evidence review methods. For implementing CAUTI prevention changes, relying first on systematic syntheses best supports standardized, evidence-based practice.
An infection preventionist is reviewing practices in a facility's food preparation department. Which of the following practices should be revised?
Options:
Thawing meat at room temperature
Using a cutting board to cut vegetables
Maintaining hot food at 145° F (62.7° C) during serving
Discarding most perishable food within 72 hours
Answer:
AExplanation:
Thawing raw meat at room temperature is a major food safety violation because it allows bacteria to multiply rapidly within the temperature danger zone (40–140°F or 4.4–60°C). Meat should always be thawed in the refrigerator, under cold running water, or in a microwave if cooked immediately.
Why the Other Options Are Incorrect?
B. Using a cutting board to cut vegetables – This is safe as long as proper cleaning and sanitation procedures are followed.
C. Maintaining hot food at 145°F (62.7°C) during serving – 145°F is an acceptable minimum temperature for certain meats like beef, fish, and pork.
D. Discarding most perishable food within 72 hours – Many perishable foods, especially leftovers, should be discarded within 3 days, making this an appropriate practice.
CBIC Infection Control Reference
The APIC guidelines emphasize that raw meat should never be thawed at room temperature due to the risk of bacterial growth and foodborne illness.
A family, including an infant of 8 months, is going on a vacation to Europe. An infection preventionist would recommend:
Options:
Exposure to rabies should be avoided.
Family members should be vaccinated for yellow fever.
The infant should not travel until at least 12 months of age.
Family immunization records should be reviewed by their provider.
Answer:
DExplanation:
When advising a family, including an 8-month-old infant, planning a vacation to Europe, an infection preventionist (IP) must consider travel-related health risks and vaccination recommendations tailored to the destination and age-specific guidelines. The Certification Board of Infection Control and Epidemiology (CBIC) emphasizes the "Education and Training" domain, which includes providing evidence-based advice to prevent infections, aligning with the Centers for Disease Control and Prevention (CDC) and World Health Organization (WHO) travel health recommendations.
Option D, "Family immunization records should be reviewed by their provider," is the most appropriate recommendation. Europe, as a region, includes countries with varying health risks, but it is generally considered a low-risk area for many vaccine-preventable diseases compared to tropical regions. The CDC’s "Travelers’ Health" guidelines (2023) recommend that all travelers, including infants, have their immunization status reviewed by a healthcare provider prior to travel to ensure compliance with routine vaccinations (e.g., measles, mumps, rubella [MMR], diphtheria, tetanus, pertussis [DTaP], and polio) and to assess any destination-specific needs. For an 8-month-old, the review would confirm that the infant has received age-appropriate vaccines (e.g., the first doses of DTaP, Hib, PCV, and IPV, typically starting at 2 months) and is on schedule for the 6- and 12-month doses. This step ensures the family’s overall protection and identifies any gaps, making it a proactive and universally applicable recommendation.
Option A, "Exposure to rabies should be avoided," is a general travel safety tip applicable to any destination where rabies is endemic (e.g., parts of Eastern Europe or rural areas with wildlife). However, rabies risk in most European countries is low, and pre-exposure vaccination is not routinely recommended for travelers unless specific high-risk activities (e.g., handling bats) are planned. The CDC advises avoiding animal bites rather than vaccinating unless indicated, making this less specific and urgent than a records review. Option B, "Family members should be vaccinated for yellow fever," is incorrect. Yellow fever is not endemic in Europe, and vaccination is not required or recommended for travel to any European country. The WHO International Health Regulations (2005) and CDC list yellow fever vaccination as mandatory only for travelers from or to certain African and South American regions, rendering this irrelevant. Option C, "The infant should not travel until at least 12 months of age," lacks a clear evidence base. While some vaccines (e.g., MMR) are typically given at 12 months, the 8-month-old can travel safely if up-to-date on age-appropriate immunizations. The CDC allows travel for infants as young as 6 weeks with medical clearance, and delaying travel to 12 months is not a standard recommendation unless specific risks (e.g., disease outbreaks) are present, which are not indicated here.
The CBIC Practice Analysis (2022) and CDC Travelers’ Health resources prioritize pre-travel health assessments, including immunization reviews, as the foundation for safe travel. Option D ensures a comprehensive approach tailored to the family’s needs, making it the best recommendation for a trip to Europe.
A patient with meningococcal meningitis experiences cardiac arrest. A nurse administers mouth-to-mouth breathing during CPR. Which of the following actions should the nurse take immediately?
Options:
Use oral chlorhexidine rinse
Quarantine from others for 5 days
Begin chemoprophylaxis within 24 hours
Monitor self and seek care if symptoms develop
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies Neisseria meningitidis as a highly transmissible organism spread through respiratory droplets and direct contact with oral secretions. Healthcare personnel who have unprotected, close exposure—such as mouth-to-mouth resuscitation—to a patient with meningococcal meningitis are considered high-risk contacts.
In this scenario, the nurse had direct exposure to respiratory secretions during CPR, which constitutes a significant risk for transmission. The Study Guide emphasizes that postexposure chemoprophylaxis is indicated as soon as possible, ideally within 24 hours of exposure, to prevent invasive meningococcal disease. Recommended prophylactic agents include rifampin, ciprofloxacin, or ceftriaxone, depending on contraindications and institutional protocols.
Option A is incorrect because chlorhexidine oral rinse does not eliminate systemic infection risk. Option B is inappropriate because quarantine is not required for exposed healthcare workers who receive appropriate prophylaxis. Option D is insufficient, as monitoring alone does not adequately reduce the risk of developing disease following high-risk exposure.
Rapid initiation of chemoprophylaxis is a critical infection prevention intervention and a high-yield CIC® exam concept. Early action protects the exposed healthcare worker and prevents secondary transmission within the healthcare setting.
Steam sterilization should be validated with which of the following organisms?
Options:
Geobacillus stearothermophilus
Staphylococcus aureus
Bacillus anthracis
Bacillus atrophaeus
Answer:
AExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) clearly states that steam sterilization (moist heat sterilization) must be validated using biological indicators containing Geobacillus stearothermophilus spores. This organism is selected because its spores are highly resistant to moist heat, making them an ideal challenge organism for assessing the effectiveness of steam sterilization processes.
Biological indicators are used to confirm that sterilization conditions—such as temperature, pressure, and exposure time—are sufficient to achieve microbial inactivation. Geobacillus stearothermophilus thrives at high temperatures and demonstrates strong resistance to steam, so if these spores are destroyed, it provides high confidence that other less-resistant microorganisms, including bacteria, viruses, and fungi, have also been eliminated.
The other options are incorrect for steam sterilization validation. Staphylococcus aureus is a vegetative bacterium and is far less resistant than bacterial spores. Bacillus anthracis is not used as a biological indicator due to safety concerns and lack of standardization. Bacillus atrophaeus is used as the biological indicator for dry heat and ethylene oxide sterilization, not steam.
Understanding which biological indicators correspond to specific sterilization modalities is a high-yield topic on the CIC® exam and is essential for ensuring compliance with evidence-based sterilization and disinfection standards.
=======
When conducting a literature search which of the following study designs may provide the best evidence of a direct causal relationship between the experimental factor and the outcome?
Options:
A case report
A descriptive study
A case control study
A randomized-controlled trial
Answer:
DExplanation:
To determine the best study design for providing evidence of a direct causal relationship between an experimental factor and an outcome, it is essential to understand the strengths and limitations of each study design listed. The goal is to identify a design that minimizes bias, controls for confounding variables, and establishes a clear cause-and-effect relationship.
A. A case report: A case report is a detailed description of a single patient or a small group of patients with a particular condition or outcome, often including the experimental factor of interest. While case reports can generate hypotheses and highlight rare occurrences, they lack a control group and are highly susceptible to bias. They do not provide evidence of causality because they are observational and anecdotal in nature. This makes them the weakest design for establishing a direct causal relationship.
B. A descriptive study: Descriptive studies, such as cross-sectional or cohort studies, describe the characteristics or outcomes of a population without manipulating variables. These studies can identify associations between an experimental factor and an outcome, but they do not establish causality due to the absence of randomization or control over confounding variables. For example, a descriptive study might show that a certain infection rate is higher in a group exposed to a specific factor, but it cannot prove the factor caused the infection without further evidence.
C. A case control study: A case control study compares individuals with a specific outcome (cases) to those without (controls) to identify factors that may contribute to the outcome. This retrospective design is useful for studying rare diseases or outcomes and can suggest associations. However, it is prone to recall bias and confounding, and it cannot definitively prove causation because the exposure is not controlled or randomized. It is stronger than case reports or descriptive studies but still falls short of establishing direct causality.
D. A randomized-controlled trial (RCT): An RCT is considered the gold standard for establishing causality in medical and scientific research. In an RCT, participants are randomly assigned to either an experimental group (exposed to the factor) or a control group (not exposed or given a placebo). Randomization minimizes selection bias and confounding variables, while the controlled environment allows researchers to isolate the effect of the experimental factor on the outcome. The ability to compare outcomes between groups under controlled conditions provides the strongest evidence of a direct causal relationship. This aligns with the principles of evidence-based practice, which the CBIC (Certification Board of Infection Control and Epidemiology) emphasizes for infection prevention and control strategies.
Based on this analysis, the randomized-controlled trial (D) is the study design that provides the best evidence of a direct causal relationship. This conclusion is consistent with the CBIC's focus on high-quality evidence to inform infection control practices, as RCTs are prioritized in the hierarchy of evidence for establishing cause-and-effect relationships.
CBIC Infection Prevention and Control (IPC) Core Competency Model (updated guidelines, 2023), which emphasizes the use of high-quality evidence, including RCTs, for validating infection control interventions.
CBIC Examination Content Outline, Domain I: Identification of Infectious Disease Processes, which underscores the importance of evidence-based study designs in infection control research.
A review of bronchoscopy specimens indicates an unusual number of Mycobacterium fortuitum–positive cultures. Which of the following observations would be the MOST likely cause of this finding?
Options:
Bronchoscopes cleaned with sporicidal solution
Inadequate cleaning prior to disinfection
Rinsing with tap water
Drying with air or alcohol
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies nontuberculous mycobacteria (NTM), including Mycobacterium fortuitum, as organisms commonly associated with water sources, particularly potable water systems. An unusual increase in M. fortuitum–positive bronchoscopy cultures is most often linked to waterborne contamination during endoscope reprocessing, making rinsing with tap water the most likely cause.
Tap water is not sterile and may harbor NTM, which are resistant to standard municipal water treatment and capable of forming biofilms within plumbing systems. If bronchoscopes are rinsed with tap water after high-level disinfection and not followed by appropriate sterile or filtered water rinses and thorough drying, organisms such as M. fortuitum may contaminate internal channels. This can lead to pseudo-outbreaks, where cultures are positive due to contamination rather than true patient infection.
Option B, inadequate cleaning prior to disinfection, can contribute to overall reprocessing failure but is less specifically associated with NTM contamination patterns. Option A is unlikely, as sporicidal solutions are effective disinfectants. Option D, drying with air or alcohol, is a recommended step to reduce microbial growth and would not cause contamination.
For CIC® exam preparation, recognizing that tap water exposure during endoscope reprocessing is a classic source of nontuberculous mycobacteria contamination is a key concept in outbreak investigation and device reprocessing surveillance.
Which of the following activities will BEST prepare a newly hired infection preventionist to present information at the facility’s orientation program?
Options:
Observing other departments’ orientation presentations
Meeting with the facility’s leadership
Reviewing principles of adult learning
Administering tuberculin skin tests to orientees
Answer:
CExplanation:
The correct answer is C, "Reviewing principles of adult learning," as this activity will best prepare a newly hired infection preventionist to present information at the facility’s orientation program. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, effective education delivery, especially for healthcare professionals during orientation, relies on understanding adult learning principles (e.g., andragogy), which emphasize learner-centered approaches, relevance to practice, and active participation. Reviewing these principles equips the infection preventionist (IP) to design and deliver content that addresses the specific needs, experiences, and motivations of the audience—such as new staff learning infection control protocols—enhancing engagement and retention (CBIC Practice Analysis, 2022, Domain IV: Education and Research, Competency 4.1 - Develop and implement educational programs). This preparation ensures the presentation is tailored, impactful, and aligned with the goal of promoting infection prevention behaviors.
Option A (observing other departments’ orientation presentations) can provide insights into presentation styles or facility norms, but it is less focused on the IP’s specific educational role and may not address the unique content of infection prevention. Option B (meeting with the facility’s leadership) is valuable for understanding organizational priorities and gaining support, but it is more about collaboration and context-setting rather than direct preparation for presenting educational material. Option D (administering tuberculin skin tests to orientees) is a clinical task related to TB screening, not a preparatory activity for designing or delivering an educational presentation.
The focus on reviewing adult learning principles aligns with CBIC’s emphasis on evidence-based education strategies to improve infection control practices among healthcare personnel (CBIC Practice Analysis, 2022, Domain IV: Education and Research, Competency 4.2 - Evaluate the effectiveness of educational programs). This approach enables the IP to effectively communicate critical information, such as hand hygiene or isolation protocols, during the orientation program.
In a retrospective case-control study, the initial case group is composed of persons
Options:
with the disease
without the disease.
with the risk factor under investigation
without the risk factor under investigation
Answer:
AExplanation:
In a retrospective case-control study, cases and controls are selected based on disease status. The case group is composed of individuals who have the disease (cases), while the control group consists of individuals without the disease. This design allows researchers to look back in time to assess exposure to potential risk factors.
Step-by-Step Justification:
Selection of Cases and Controls:
Cases: Individuals who already have the disease.
Controls: Individuals without the disease but similar in other aspects.
Direction of Study:
A retrospective study moves backward from the disease outcome to investigate potential causes or risk factors.
Data Collection:
Uses past medical records, interviews, and laboratory results to determine past exposures.
Common Use:
Useful for studying rare diseases since cases have already occurred, making it cost-effective compared to cohort studies.
Why Other Options Are Incorrect:
B. without the disease: (Incorrect) This describes the control group, not the case group.
C. with the risk factor under investigation: (Incorrect) Risk factors are identified after selecting cases and controls.
D. without the risk factor under investigation: (Incorrect) The study investigates whether cases had prior exposure, not whether they lacked a risk factor.
CBIC Infection Control References:
APIC Text, Chapter on Epidemiologic Study Design.
An infection preventionist (IP) notices that several discharged newborns have been readmitted with staphylococcal infections. What should the IP do FIRST?
Options:
Observe medical and nursing techniques in the nursery.
Obtain surveillance cultures on babies in the nursery.
Begin prospective surveillance to identify new staphylococcal cases.
Review medical records of the readmitted cases.
Answer:
DExplanation:
The Certification Study Guide (6th edition) outlines a structured approach to outbreak investigation, emphasizing that the first step is to verify the problem and establish baseline facts before initiating control measures. When an infection preventionist becomes aware of potential clustering—such as multiple newborn readmissions with staphylococcal infections—the initial priority is to review the medical records of the affected cases.
Reviewing records allows the IP to confirm diagnoses, identify common organisms, determine timing of symptom onset, and assess potential epidemiologic links (e.g., same nursery, staff exposure, procedures, or length of stay). This step helps determine whether the cases represent a true outbreak, coincidental community-acquired infections, or unrelated events. The study guide stresses that interventions should not begin until the problem is clearly defined, as premature actions may waste resources or obscure the true source.
The other options are appropriate later steps in an investigation. Observing practices and obtaining surveillance cultures are targeted control measures that should follow confirmation of an outbreak and hypothesis generation. Beginning prospective surveillance is also important, but only after case definitions and baseline data are established.
CIC exam questions frequently test sequencing of outbreak investigation steps. Recognizing that case confirmation and record review come first is essential for effective infection prevention decision-making and accurate epidemiologic analysis.
Which of the following processes is essential for endoscope reprocessing?
Options:
Intermediate level disinfection and contact time
Pre-cleaning, leak testing, and manual cleaning
Inspection using a borescope and horizontal storage
Leak testing, manual cleaning, and low level disinfection
Answer:
BExplanation:
The correct answer is B, "Pre-cleaning, leak testing, and manual cleaning," as these processes are essential for endoscope reprocessing. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, proper reprocessing of endoscopes is critical to prevent healthcare-associated infections (HAIs), given their complex design and susceptibility to microbial contamination. The initial steps of pre-cleaning (removing gross debris at the point of use), leak testing (ensuring the endoscope’s integrity to prevent fluid ingress), and manual cleaning (using enzymatic detergents to remove organic material) are foundational to the reprocessing cycle. These steps prepare the endoscope for high-level disinfection or sterilization by reducing bioburden and preventing damage, as outlined in standards such as AAMI ST91 (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.3 - Ensure safe reprocessing of medical equipment). Failure at this stage can compromise subsequent disinfection, making it a non-negotiable component of the process.
Option A (intermediate level disinfection and contact time) is an important step but insufficient alone, as intermediate-level disinfection does not achieve the high-level disinfection required for semi-critical devices like endoscopes, which must eliminate all microorganisms except high levels of bacterial spores. Option C (inspection using a borescope and horizontal storage) includes valuable quality control (inspection) and storage practices, but these occur later in the process and are not essential initial steps; vertical storage is often preferred to prevent damage. Option D (leak testing, manual cleaning, and low level disinfection) includes two essential steps (leak testing and manual cleaning) but is inadequate because low-level disinfection does not meet the standard for endoscopes, which require high-level disinfection or sterilization.
The emphasis on pre-cleaning, leak testing, and manual cleaning aligns with CBIC’s focus on adhering to evidence-based reprocessing protocols to ensure patient safety and prevent HAIs (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.4 - Implement environmental cleaning and disinfection protocols). These steps are mandated by guidelines to mitigate risks associated with endoscope use in healthcare settings.
Immediate use steam sterilization is NOT recommended for implantable items requiring immediate use because
Options:
the high temperature may damage the items.
chemical indicators may not be accurate at high temperatures.
results of biologic indicators are unavailable prior to use of the item.
the length of time is inadequate for the steam to penetrate the pack.
Answer:
CExplanation:
The correct answer is C, "results of biologic indicators are unavailable prior to use of the item," as this is the primary reason immediate use steam sterilization (IUSS) is not recommended for implantable items requiring immediate use. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, IUSS is a process used for sterilizing items needed urgently when no other sterile options are available, typically involving a shortened cycle (e.g., flash sterilization). However, for implantable items—such as orthopedic hardware or prosthetic devices—ensuring absolute sterility is critical due to the risk of deep infection. Biologic indicators (BIs), which contain highly resistant spores to verify sterilization efficacy, require incubation (typically 24-48 hours) to confirm the kill, but IUSS does not allow time for BI results to be available before the item is used (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.3 - Ensure safe reprocessing of medical equipment). This lack of immediate verification poses a significant infection risk, making IUSS inappropriate for implants, as per AAMI ST79 standards.
Option A (the high temperature may damage the items) is a consideration for some heat-sensitive materials, but modern IUSS cycles are designed to minimize damage, and this is not the primary reason for the restriction on implants. Option B (chemical indicators may not be accurate at high temperatures) is incorrect, as chemical indicators (e.g., color-changing strips) are reliable at high temperatures and serve as an immediate check, though they are not a substitute for BIs. Option D (the length of time is inadequate for the steam to penetrate the pack) is not the main issue, as IUSS cycles are optimized for penetration, though the shortened time may be a secondary concern; the unavailability of BI results remains the decisive factor.
The focus on biologic indicator results aligns with CBIC’s emphasis on ensuring the safety and sterility of reprocessed medical devices, particularly for high-risk implantable items (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.5 - Evaluate the environment for infection risks). This recommendation is supported by AAMI and CDC guidelines, which prioritize BI confirmation for implants to prevent healthcare-associated infections (AAMI ST79:2017, CDC Sterilization Guidelines, 2019).
A 17-year-old presents to the Emergency Department with fever, stiff neck, and vomiting. A lumbar puncture is done. The Gram stain shows Gram negative diplocooci. Presumptive identification of the organism is
Options:
Haemophilus influenzae
Neisseria meningitidis
Listeria monocytogenes
Streptococcus pneumoniae
Answer:
BExplanation:
The Gram stain showing Gram-negative diplococci in cerebrospinal fluid (CSF) is characteristic of Neisseria meningitidis, a leading cause of bacterial meningitis in adolescents and young adults.
Step-by-Step Justification:
Gram Stain Interpretation:
Gram-negative diplococci in CSF strongly suggest Neisseria meningitidis.
Classic Symptoms of Meningitis:
Fever, stiff neck, and vomiting are hallmark signs of meningococcal meningitis.
Neisseria meningitidis vs. Other Bacteria:
Haemophilus influenzae (Option A) → Gram-negative coccobacilli.
Listeria monocytogenes (Option C) → Gram-positive rods.
Streptococcus pneumoniae (Option D) → Gram-positive diplococci.
CBIC Infection Control References:
APIC Ready Reference for Microbes, "Neisseria meningitidis and Meningitis".
Major construction and renovations are planned for a hospital’s operating suite, and a meeting is scheduled to plan for construction activities. Aside from the infection preventionist, and representatives from environmental services and engineering, who else should be included in these planning conversations?
Options:
Operating room nurse manager
Chief operating officer
Plumbing supervisor
Director of public relations
Answer:
AExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that multidisciplinary collaboration is essential when planning construction or renovation projects in patient care areas, especially high-risk locations such as operating suites. In addition to infection prevention, environmental services, and engineering, the operating room nurse manager must be actively involved in construction planning discussions.
The operating room nurse manager represents frontline clinical operations and has direct knowledge of surgical workflows, patient movement, sterile processing needs, case scheduling, and staff practices. Their involvement ensures that construction activities are coordinated to minimize disruption to patient care, maintain sterile environments, and reduce infection risks associated with dust, airflow changes, and traffic patterns. The nurse manager also plays a key role in communicating construction-related precautions and practice changes to surgical staff.
While senior leadership (Option B) may provide oversight, they are not typically involved in detailed infection control planning. The plumbing supervisor (Option C) may be consulted for specific infrastructure issues but does not represent clinical operations. The director of public relations (Option D) is not relevant to construction-related infection risk planning.
The Study Guide highlights that ICRA planning must include clinical leadership from affected areas to ensure that infection prevention measures are practical, effective, and consistently implemented. Including the operating room nurse manager is therefore essential for safe construction planning and is a frequently tested CIC® exam concept.
==========
The infection preventionist (IP) is notified about the recall of certain ice machines because of a faulty filtration device. These ice machines are located on several units throughout the facility. What is the BEST action the IP should take?
Options:
Culture all ice machines for Legionella
Report a waterborne disease outbreak to Administration
Identify all patients who have been served ice from these machines
Supply an alternative source of ice while investigating further
Answer:
DExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that when a device recall involves potential contamination risk, the infection preventionist’s first priority is risk mitigation and prevention of further exposure. In this scenario, the recall of ice machines due to a faulty filtration device represents a potential waterborne contamination risk, even in the absence of confirmed infections.
The best immediate action is to remove the recalled ice machines from service and provide an alternative source of ice while further investigation and corrective actions are underway. This step promptly eliminates the exposure pathway and protects patients, staff, and visitors from possible contamination. The Study Guide stresses that interruption of use is the most effective initial control measure when equipment safety is in question.
Option A is incorrect because culturing ice machines is not the first step and is not routinely recommended without clinical indication. Option B is inappropriate because there is no evidence of a confirmed outbreak. Option C may be necessary later if exposure investigation becomes warranted, but it should not precede immediate risk control.
For the CIC® exam, it is essential to recognize that eliminating exposure takes precedence over testing or notification activities. Supplying an alternative ice source while investigating further aligns with risk management principles, patient safety priorities, and evidence-based infection prevention practice.
==========
Properly written instructional objectives should:
Options:
Communicate the intent of the program.
Describe learner outcomes using action words.
Determine whether or not continuing education units may be offered.
Be limited to the knowledge and application levels of Bloom’s taxonomy.
Answer:
BExplanation:
Properly written instructional objectives are a fundamental component of effective education programs and are emphasized in the Education and Research domain of the CBIC Certified Infection Control Exam Study Guide (6th edition). Instructional objectives are designed to clearly state what the learner will be able to do after completing an educational activity. The Study Guide highlights that objectives must be learner-centered, measurable, and observable, which is best achieved by using clear action-oriented verbs.
Describing learner outcomes using action words—such as identify, analyze, demonstrate, apply, or evaluate—allows educators to define expected performance and assess whether learning has occurred. These action words are typically aligned with Bloom’s taxonomy and support evaluation of cognitive, psychomotor, or affective learning domains. This approach ensures that education is outcome-driven rather than content-driven.
Option A is incorrect because communicating the intent of the program is the purpose of a program goal, not an instructional objective. Option C is unrelated to instructional design; continuing education unit eligibility is determined by accrediting bodies, not by objectives themselves. Option D is incorrect because instructional objectives are not limited to knowledge and application levels; they may address higher-order thinking skills such as analysis, synthesis, and evaluation.
For CIC® exam preparation, recognizing that instructional objectives must be written in measurable, action-oriented terms is essential, as this principle directly supports effective education, competency validation, and performance improvement in infection prevention programs.
==========
aQUESTION NO: 5
Following an aerosol release of anthrax, a hospital distributes antibiotic prophylaxis to all of its employees and their family members but not to members of the general public. What is the hospital implementing?
A. Closed point of dispensing
B. Hospital incident command
C. Occupational health policy
D. Syndromic surveillance
Answer: A
In the context of a biologic emergency such as an aerosolized release of anthrax, rapid distribution of prophylactic medications is a critical preparedness function. The CBIC Certified Infection Control Exam Study Guide (6th edition) describes a closed point of dispensing (POD) as a mechanism by which an organization dispenses medications or vaccines to a defined, non-public population, such as employees and their families, rather than the general public.
Hospitals commonly serve as closed PODs during public health emergencies to ensure continuity of operations. By providing antibiotic prophylaxis to healthcare workers and their household contacts, the hospital reduces absenteeism, protects its workforce, and maintains its ability to deliver patient care during a crisis. This approach is typically coordinated with public health authorities but is operationally managed by the organization for its designated population.
The other options do not best fit the scenario. Hospital incident command is a management structure used to coordinate response activities but does not specifically describe medication distribution. An occupational health policy governs routine employee health practices and does not extend to family members during emergency prophylaxis. Syndromic surveillance refers to monitoring data for early detection of outbreaks, not to dispensing antibiotics.
Closed POD operations are a key component of emergency preparedness and bioterrorism response planning, and recognition of this concept is essential for CIC® exam candidates.
==========
An infection preventionist is developing training exercises for emergency preparedness and disaster response teams. The MOST effective instructional method for retaining information is:
Options:
Providing reading materials to the group.
Watching videos recorded by other hospitals.
Simulating an event to practice how to respond.
Administering a post-test after circulating the emergency response plan.
Answer:
CExplanation:
The Certification Study Guide (6th edition) emphasizes that active, experiential learning methods are the most effective for long-term retention of knowledge and skills, particularly in the context of emergency preparedness and disaster response. Simulation-based training allows participants to practice real-time decision-making, communication, and task execution in a controlled environment that closely mirrors actual emergency conditions.
Simulating an event—such as a mass casualty incident, infectious disease outbreak, or evacuation—engages learners cognitively, physically, and emotionally. The study guide notes that this type of hands-on training improves recall, reinforces correct behaviors, exposes system gaps, and builds team confidence. Simulation also supports interdisciplinary coordination and allows immediate feedback and debriefing, which further enhances learning retention.
The other instructional methods are less effective for retention. Reading materials and watching videos are passive learning approaches that may increase awareness but do not ensure competency during high-stress situations. Administering a post-test measures short-term knowledge acquisition but does not demonstrate the ability to apply that knowledge during an actual emergency.
CIC exam questions frequently highlight adult learning principles, stressing that people learn best by doing—especially when preparing for rare but high-risk events. Simulation-based exercises are therefore considered the gold standard for emergency preparedness training and are strongly recommended for disaster response teams.
Which statistical test is MOST appropriate for comparing infection rates before and after an intervention?
Options:
Student’s t-test
Chi-square test for proportions
Linear regression analysis
Wilcoxon rank-sum test
Answer:
BExplanation:
The Chi-square test is the most appropriate test for comparing infection rates (categorical data) before and after an intervention.
CBIC Infection Control References:
CIC Study Guide, "Statistical Analysis in Infection Control," Chapter 5.
Assume the mean age of onset for patients with tuberculosis (TB) is 62 years, with one standard deviation of 5 years, and the age of onset follows a normal distribution. What is the percentage of patients expected to have the age of onset ranging from 57 to 67 years?
Options:
34%
68%
95%
99%
Answer:
BExplanation:
To determine the percentage of patients with an age of onset ranging from 57 to 67 years, we need to apply the properties of a normal distribution. In a normal distribution, the mean represents the central point, and the standard deviation defines the spread of the data. Here, the mean age of onset is 62 years, and the standard deviation is 5 years. The range of 57 to 67 years corresponds to one standard deviation below the mean (62 - 5 = 57) to one standard deviation above the mean (62 + 5 = 67).
In a normal distribution, approximately 68% of the data falls within one standard deviation of the mean (i.e., between μ - σ and μ + σ, where μ is the mean and σ is the standard deviation). This is a well-established statistical principle, often referred to as the 68-95-99.7 rule (or empirical rule) in statistics. Specifically, 34% of the data lies between the mean and one standard deviation above the mean, and another 34% lies between the mean and one standard deviation below the mean, totaling 68% for the range spanning one standard deviation on both sides of the mean.
Let’s verify this:
The lower bound (57 years) is exactly one standard deviation below the mean (62 - 5 = 57).
The upper bound (67 years) is exactly one standard deviation above the mean (62 + 5 = 67).
Thus, the range from 57 to 67 years encompasses the middle 68% of the distribution.
Option A (34%) represents the percentage of patients within one standard deviation on only one side of the mean (e.g., 62 to 67 or 57 to 62), not the full range. Option C (95%) corresponds to approximately two standard deviations from the mean (62 ± 10 years, or 52 to 72 years), which is wider than the given range. Option D (99%) aligns with approximately three standard deviations (62 ± 15 years, or 47 to 77 years), which is even broader. Since the question specifies a range of one standard deviation on either side of the mean, the correct answer is 68%, corresponding to Option B.
In infection control, understanding the distribution of disease onset ages can help infection preventionists identify at-risk populations and allocate resources effectively, aligning with the CBIC’s focus on surveillance and data analysis (CBIC Practice Analysis, 2022). While the CBIC does not directly address statistical calculations in its core documents, the application of normal distribution principles is a standard epidemiological tool endorsed in public health guidelines, which inform CBIC practices.
Documentation of each steam sterilization cycle should include which of the following pieces of information?
Options:
Load contents
Machine model number
Date sterilizer was cleaned
Initials of the person who prepared the instrument set
Answer:
AExplanation:
Documentation of each steam sterilization cycle is a regulatory and quality requirement. It must include load contents, the sterilizer ID, date, cycle number, and the person who assembled the load. These details support traceability and quality assurance.
The APIC Text states:
“Each item or package should be labeled with a lot-control identifier that includes the sterilizer identification number or code, a detailed list of the contents, an identifier for the person who assembled the package, the date of sterilization, the cycle number...”
Other options like the machine model number or date sterilizer was cleaned are not routine documentation elements for every cycle.
An environmental services staff member sustained a needle-stick injury while collecting garbage and immediately called the Occupational Health (OH) department for further direction. What should the OH department recommend FIRST?
Options:
Administer first aid treatment
Schedule an appointment with the OH department
Discuss the exposure incident with staff to decide on the level of risk
Obtain and assess immune status to bloodborne pathogens
Answer:
AExplanation:
The Certification Study Guide (6th edition) emphasizes that immediate first aid is the first and most critical step following an occupational exposure to blood or body fluids, including needle-stick injuries. First aid measures include promptly washing the affected area with soap and water and flushing mucous membranes with water if exposed. This immediate action helps reduce the microbial load at the exposure site and may lower the risk of transmission of bloodborne pathogens such as hepatitis B virus (HBV), hepatitis C virus (HCV), and human immunodeficiency virus (HIV).
The study guide outlines a clear sequence for managing occupational exposures. Initial wound care always precedes risk assessment, documentation, immune status evaluation, and post-exposure prophylaxis decisions. Delaying first aid to gather information or schedule appointments is inconsistent with best practice and increases potential risk to the exposed worker.
The other options represent appropriate subsequent steps, not first actions. Scheduling an OH appointment and assessing immune status are important but occur after immediate wound care. Discussing the exposure to determine risk level is also essential, but only after first aid has been provided.
CIC exam questions frequently assess understanding of prioritization and sequencing in occupational exposure management. Recognizing that immediate first aid is always the first intervention reflects sound infection prevention practice and aligns with established occupational health protocols.
When a Staphylococcus aureus outbreak is suspected, cultures of which of the following sites will MOST likely identify carriers?
Options:
Nose
Hands
Throat
Rectum
Answer:
AExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies the anterior nares (nose) as the most common and reliable site for colonization with Staphylococcus aureus, including methicillin-resistant Staphylococcus aureus (MRSA). During suspected outbreaks, culturing the nares is the most effective method for identifying persistent carriers, particularly among healthcare personnel or patients who may serve as reservoirs for transmission.
Nasal carriage of S. aureus is well established in epidemiologic literature and infection prevention practice. Individuals may be persistent carriers, intermittent carriers, or non-carriers, with persistent nasal carriers posing the highest risk for transmission and subsequent infection. The Study Guide emphasizes that nasal colonization strongly correlates with both endogenous infection risk and spread to others, making it the preferred screening site during outbreak investigations.
Hands (Option B) may transiently harbor S. aureus, but hand contamination is temporary and highly variable, making it less useful for identifying long-term carriers. Throat (Option C) and rectum (Option D) are not primary colonization sites for S. aureus and are not routinely used in outbreak screening unless specifically indicated by epidemiologic data.
For CIC® exam purposes, this question reinforces a core infection prevention principle: the anterior nares are the primary reservoir for Staphylococcus aureus, and nasal cultures are the most effective method for identifying carriers during outbreak investigations.
An outbreak of carbapenem-resistant Klebsiella pneumoniae is linked to duodenoscopes. What is the infection preventionist’s PRIORITY intervention?
Options:
Perform targeted patient screening for Klebsiella pneumoniae.
Implement immediate enhanced reprocessing procedures and audit compliance.
Discontinue the use of duodenoscopes until further notice.
Conduct whole-genome sequencing of outbreak isolates.
Answer:
BExplanation:
The CDC and FDA have identified duodenoscopes as high-risk devices due to inadequate reprocessing, leading to MDRO transmission.
The first priority is enhancing reprocessing protocols and ensuring strict compliance with manufacturer instructions.
CBIC Infection Control References:
APIC Text, "Endoscope Reprocessing and Infection Risk," Chapter 10.
A director of the Infection Prevention and Control Department has received permission to hire more staff to a total of 3.8 full-time equivalents (FTEs), based on 40 hours/week. The director works 40 hours/week, one infection preventionist (IP) works 32 hours/week, another works 16 hours/week, and secretarial support works 40 hours/week. How many additional FTEs may the director hire?
Options:
0.4
0.6
0.8
1.0
Answer:
BExplanation:
The Certification Study Guide (6th edition) stresses that infection prevention leaders must understand basic workforce and FTE calculations to ensure appropriate staffing and compliance with approved resource allocations. An FTE is defined as 40 hours worked per week, and part-time hours must be converted proportionally.
First, calculate the FTEs already in use:
Director: 40 hours/week ÷ 40 = 1.0 FTE
Infection preventionist: 32 hours/week ÷ 40 = 0.8 FTE
Infection preventionist: 16 hours/week ÷ 40 = 0.4 FTE
Secretarial support: 40 hours/week ÷ 40 = 1.0 FTE
Total current FTEs:
1.0 + 0.8 + 0.4 + 1.0 = 3.2 FTEs
The approved staffing total is 3.8 FTEs. To determine how many additional FTEs may be hired, subtract current FTE usage from the approved total:
3.8 − 3.2 = 0.6 FTE
Therefore, the director may hire 0.6 additional FTE, which could be fulfilled by a part-time infection preventionist or split among staff roles, depending on organizational needs.
CIC exam questions frequently test practical management skills, including staffing calculations, budgeting awareness, and resource allocation. Accurate FTE calculations ensure compliance with administrative approvals and support safe, effective infection prevention program operations.
An infection preventionist (IP) observes an increase in primary bloodstream infections in patients admitted through the Emergency Department. Poor technique is suspected when peripheral intravenous (IV) catheters are inserted. The IP should FIRST stratify infections by:
Options:
Location of IV insertion: pre-hospital, Emergency Department, or in-patient unit.
Type of dressing used: gauze, CHG impregnated sponge, or transparent.
Site of insertion: hand, forearm, or antecubital fossa.
Type of skin preparation used for the IV site: alcohol, CHG/alcohol, or iodophor.
Answer:
AExplanation:
When an infection preventionist (IP) identifies an increase in primary bloodstream infections (BSIs) associated with peripheral intravenous (IV) catheter insertion, the initial step in outbreak investigation and process improvement is to stratify the data to identify potential sources or patterns of infection. According to the Certification Board of Infection Control and Epidemiology (CBIC), the "Surveillance and Epidemiologic Investigation" domain emphasizes the importance of systematically analyzing data to pinpoint contributing factors, such as location, technique, or equipment use, in healthcare-associated infections (HAIs). The question specifies poor technique as a suspected cause, and the first step should focus on contextual factors that could influence technique variability.
Option A, stratifying infections by the location of IV insertion (pre-hospital, Emergency Department, or in-patient unit), is the most logical first step. Different settings may involve varying levels of training, staffing, time pressure, or adherence to aseptic technique, all of which can impact infection rates. For example, pre-hospital settings (e.g., ambulance services) may have less controlled environments or less experienced personnel compared to in-patient units, potentially leading to technique inconsistencies. The CDC’s Guidelines for the Prevention of Intravascular Catheter-Related Infections (2017) recommend evaluating the context of catheter insertion as a critical initial step in investigating BSIs, making this a priority for the IP to identify where the issue is most prevalent.
Option B, stratifying by the type of dressing used (gauze, CHG impregnated sponge, or transparent), is important but should follow initial location-based analysis. Dressings play a role in maintaining catheter site integrity and preventing infection, but their impact is secondary to the insertion technique itself. Option C, stratifying by the site of insertion (hand, forearm, or antecubital fossa), is also relevant, as anatomical sites differ in infection risk (e.g., the hand may be more prone to contamination), but this is a more specific factor to explore after broader contextual data is assessed. Option D, stratifying by the type of skin preparation used (alcohol, CHG/alcohol, or iodophor), addresses antiseptic efficacy, which is a key component of technique. However, without first understanding where the insertions occur, it’s premature to focus on skin preparation alone, as technique issues may stem from systemic factors across locations.
The CBIC Practice Analysis (2022) supports a stepwise approach to HAI investigation, starting with broad stratification (e.g., by location) to guide subsequent detailed analysis (e.g., technique-specific factors). This aligns with the CDC’s hierarchical approach to infection prevention, where contextual data collection precedes granular process evaluation. Therefore, the IP should first stratify by location to establish a baseline for further investigation.
Which of the following blood-drawing methods is considered to be a needle-safe practice?
Options:
Use a syringe with a needle attached.
Shielded needles for vacuum-tube phlebotomy sets.
Remove contaminated needles from blood collection sets.
Inject blood into vacuum tubes using conventional syringes.
Answer:
BExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies engineered sharps injury prevention devices (ESIPDs) as the cornerstone of needle-safe practices during blood collection. Shielded needles used with vacuum-tube phlebotomy systems are specifically designed to reduce the risk of needlestick injuries by incorporating a built-in safety mechanism that covers or retracts the needle immediately after use.
Vacuum-tube systems with shielded needles allow blood to flow directly into collection tubes without the need for needle removal or blood transfer, thereby minimizing handling of sharps. Once blood collection is complete, the safety feature is activated—often automatically or with a single-handed technique—significantly reducing exposure risk to healthcare personnel. The Study Guide emphasizes that these devices meet regulatory expectations under the Needlestick Safety and Prevention Act and should be used whenever feasible.
The other options are unsafe practices. Using syringes with attached needles (Option A) increases risk during transfer and disposal. Removing contaminated needles from collection sets (Option C) is explicitly prohibited due to high injury risk. Injecting blood into vacuum tubes using conventional syringes (Option D) requires manipulating exposed needles and increases the likelihood of splashes and sharps injuries.
For CIC® exam preparation, it is essential to recognize that needle-safe blood collection relies on safety-engineered devices, with shielded vacuum-tube phlebotomy needles representing best practice for preventing occupational exposures.
An infection preventionist reviewing patient records in an outpatient hemodialysis center notes an increase in localized infections at catheter access sites. Which of the following strategies reduces the risk of infection in this population?
Options:
Creation of an arteriovenous fistula
Use of a non-cuffed percutaneous catheter
Placement of a femoral catheter
Replacement of dialysis catheters monthly
Answer:
AExplanation:
The best strategy to reduce the risk of infection in hemodialysis patients is to use an arteriovenous (AV) fistula as the preferred vascular access method. AV fistulas have the lowest infection rates compared to catheters and grafts because they do not involve foreign material and are less prone to biofilm formation and bloodstream infections.
Why the Other Options Are Incorrect?
B. Use of a non-cuffed percutaneous catheter – Non-cuffed catheters have a higher risk of bloodstream infections and should be used only for short-term access.
C. Placement of a femoral catheter – Femoral catheters have higher infection risks and should only be used for bed-bound patients and for the shortest duration possible.
D. Replacement of dialysis catheters monthly – Routine catheter replacement does not reduce infection risk and should be done only when medically necessary.
CBIC Infection Control Reference
According to APIC guidelines, AV fistulas are the preferred vascular access due to their lower infection rates and improved long-term outcomes.
A new hospital disinfectant with a 3-minute contact time has been purchased by Environmental Services. The disinfectant will be rolled out across the patient care 3-minute contact time has been purchased by Environmental Services. The disinfectant will be rolled out across the patient care areas. They are concerned about the high cost of the disinfectant. What advice can the infection preventionist provide?
Options:
Use the new disinfectant for patient washrooms only.
Use detergents on the floors in patient rooms.
Use detergents on smooth horizontal surfaces.
Use new disinfectant for all surfaces in the patient room.
Answer:
CExplanation:
The scenario involves the introduction of a new hospital disinfectant with a 3-minute contact time, intended for use across patient care areas, but with concerns raised by Environmental Services about its high cost. The infection preventionist’s advice must balance infection control efficacy with cost management, adhering to principles outlined by the Certification Board of Infection Control and Epidemiology (CBIC) and evidence-based practices. The goal is to optimize the disinfectant’s use while ensuring a safe environment. Let’s evaluate each option:
A. Use the new disinfectant for patient washrooms only: Limiting the disinfectant to patient washrooms focuses its use on high-touch, high-risk areas where pathogens (e.g., Clostridioides difficile, norovirus) may be prevalent. However, this approach restricts the disinfectant’s application to a specific area, potentially leaving other patient care surfaces (e.g., bed rails, tables) vulnerable to contamination. While cost-saving, it does not address the broad infection control needs across all patient care areas, making it an incomplete strategy.
B. Use detergents on the floors in patient rooms: Detergents are cleaning agents that remove dirt and organic material but lack the antimicrobial properties of disinfectants. Floors in patient rooms can harbor pathogens, but they are generally considered lower-risk surfaces compared to high-touch areas (e.g., bed rails, doorknobs). Using detergents instead of the new disinfectant on floors could reduce costs but compromises infection control, as floors may still contribute to environmental transmission (e.g., via shoes or equipment). This option is not optimal given the availability of an effective disinfectant.
C. Use detergents on smooth horizontal surfaces: Smooth horizontal surfaces (e.g., tables, counters, overbed tables) are common sites for pathogen accumulation and transmission in patient rooms. Using detergents to clean these surfaces removes organic material, which is a critical first step before disinfection. If the 3-minute contact time disinfectant is reserved for high-touch or high-risk surfaces (e.g., bed rails, call buttons) where disinfection is most critical, this approach maximizes the disinfectant’s efficacy while reducing its overall use and cost. This strategy aligns with CBIC guidelines, which emphasize a two-step process (cleaning followed by disinfection) and targeted use of resources, making it a practical and cost-effective recommendation.
D. Use new disinfectant for all surfaces in the patient room: Using the disinfectant on all surfaces ensures comprehensive pathogen reduction but increases consumption and cost, which is a concern for Environmental Services. While the 3-minute contact time suggests efficiency, overusing the disinfectant on low-risk surfaces (e.g., floors, walls) may not provide proportional infection control benefits and could strain the budget. This approach does not address the cost concern and is less strategic than targeting high-risk areas.
The best advice is C, using detergents on smooth horizontal surfaces to handle routine cleaning, while reserving the new disinfectant for high-touch or high-risk areas where its antimicrobial action is most needed. This optimizes infection prevention, aligns with CBIC’s emphasis on evidence-based environmental cleaning, and addresses the cost concern by reducing unnecessary disinfectant use. The infection preventionist should also recommend a risk assessment to identify priority surfaces for disinfectant application.
CBIC Infection Prevention and Control (IPC) Core Competency Model (updated 2023), Domain IV: Environment of Care, which advocates for targeted cleaning and disinfection based on risk.
CBIC Examination Content Outline, Domain III: Prevention and Control of Infectious Diseases, which includes cost-effective use of disinfectants.
CDC Guidelines for Environmental Infection Control in Healthcare Facilities (2022), which recommend cleaning with detergents followed by targeted disinfection.
An HBsAb-negative employee has a percutaneous exposure to blood from a Hepatitis B surface antigen (HBsAg) positive patient. Which of the following regimens is recommended for this employee?
Options:
Immune serum globulin and hepatitis B vaccine
Hepatitis B immune globulin (HBIG) alone
Hepatitis B vaccine alone
Hepatitis B immune globulin (HBIG) and hepatitis B vaccine
Answer:
DExplanation:
The correct answer is D, "Hepatitis B immune globulin (HBIG) and hepatitis B vaccine," as this is the recommended regimen for an HBsAb-negative employee with a percutaneous exposure to blood from an HBsAg-positive patient. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, which align with recommendations from the Centers for Disease Control and Prevention (CDC) and the Advisory Committee on Immunization Practices (ACIP), post-exposure prophylaxis (PEP) for hepatitis B virus (HBV) exposure depends on the employee’s vaccination status and the source’s HBsAg status. For an unvaccinated or known HBsAb-negative individual (indicating no immunity) exposed to HBsAg-positive blood, the standard PEP includes both HBIG and the hepatitis B vaccine. HBIG provides immediate passive immunity by delivering pre-formed antibodies, while the vaccine initiates active immunity to prevent future infections (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.2 - Implement measures to prevent transmission of infectious agents). The HBIG should be administered within 24 hours of exposure (preferably within 7 days), and the first dose of the vaccine should be given concurrently, followed by the complete vaccine series.
Option A (immune serum globulin and hepatitis B vaccine) is incorrect because immune serum globulin (ISG) is a general immunoglobulin preparation and not specific for HBV; HBIG, which contains high titers of anti-HBs, is the appropriate specific immunoglobulin for HBV exposure. Option B (hepatitis B immune globulin [HBIG] alone) is insufficient, as it provides only temporary passive immunity without initiating long-term active immunity through vaccination, which is critical for an unvaccinated individual. Option C (hepatitis B vaccine alone) is inadequate for immediate post-exposure protection, as it takes weeks to develop immunity, leaving the employee vulnerable in the interim.
The recommendation for HBIG and hepatitis B vaccine aligns with CBIC’s emphasis on evidence-based post-exposure management to prevent HBV transmission in healthcare settings (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.1 - Collaborate with organizational leaders). This dual approach is supported by CDC guidelines, which prioritize rapid intervention to reduce the risk of seroconversion following percutaneous exposure (CDC Updated U.S. Public Health Service Guidelines for the Management of Occupational Exposures to HBV, HCV, and HIV, 2013).
To understand how their hospital-acquired infection rates compare to other health care settings, an infection preventionist (IP) plans to use benchmarking.
Which of the following criteria is important to ensure accurate benchmarking of surveillance data?
Options:
Data collectors are trained on how to collect data
Collecting data on a small population lo ensure accuracy of data collection
Denominator rates are selected based on an organizational risk assessment
Using case definitions that are adjusted for the patient population being studied
Answer:
DExplanation:
Benchmarking compares infection rates across healthcare facilities. For accurate benchmarking, case definitions must be standardized and adjusted for patient demographics, severity of illness, and other risk factors.
Why the Other Options Are Incorrect?
A. Data collectors are trained on how to collect data – Training is necessary, but it does not directly ensure comparability between facilities.
B. Collecting data on a small population – A larger sample size increases accuracy and reliability in benchmarking.
C. Denominator rates selected based on an organizational risk assessment – Risk assessment is important, but standardized case definitions are critical for comparison.
CBIC Infection Control Reference
According to APIC, accurate benchmarking relies on using standardized case definitions that account for differences in patient populations.
Which of the following measures has NOT been demonstrated to reduce the risk of surgical site infections?
Options:
Limiting the duration of preoperative hospital stay
Using antimicrobial preoperative scrub by members of the surgical team
Assuring adequate patient nutrition
Designating a specific surgical suite tor infected cases
Answer:
DExplanation:
There is no strong evidence that isolating infected cases in a separate surgical suite reduces SSI risk.
Step-by-Step Justification:
SSI Prevention Strategies Supported by Evidence:
Preoperative hospital stay limitation reduces exposure to hospital-acquired pathogens.
Antimicrobial preoperative scrubs lower bacterial load on the skin.
Adequate nutrition improves immune function and wound healing.
Why Designating a Separate Surgical Suite Is Not Effective:
Operating room environmental controls (e.g., laminar airflow, sterilization protocols) are more important than suite designation.
No significant reduction in SSIs has been observed by segregating infected cases into specific OR suites.
Why Other Options Are Correct:
A. Limiting preoperative hospital stay: Reduces nosocomial bacterial exposure.
B. Antimicrobial preoperative scrub: Decreases skin flora contamination.
C. Assuring adequate patient nutrition: Enhances immune defense against infections.
CBIC Infection Control References:
APIC Text, "Surgical Site Infection Prevention Strategies".
In the current year, cases of tuberculosis (TB) among foreign-born persons accounted for the majority of new TB cases in the United States. The number of states with greater than 50% of cases among foreign-born persons increased from four cases ten years ago to 22 cases in the current year. This information can BEST be used to
heighten awareness among Emergency Department staff.
inform staff who are foreign-born.
educate patients and visitors.
review the TB exposure control plan.
Options:
1 and 2 only.
1 and 4 only.
2 and 3 only.
3 and 4 only.
Answer:
BExplanation:
The correct answer is B, "1 and 4 only," indicating that the information can best be used to heighten awareness among Emergency Department (ED) staff and review the TB exposure control plan. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, tuberculosis (TB) remains a significant public health concern, particularly with the increasing proportion of cases among foreign-born persons in the United States. The data showing a rise from four to 22 states with over 50% of TB cases among foreign-born individuals highlights an evolving epidemiological trend that warrants targeted infection prevention strategies (CBIC Practice Analysis, 2022, Domain II: Surveillance and Epidemiologic Investigation, Competency 2.1 - Conduct surveillance for healthcare-associated infections and epidemiologically significant organisms).
Heightening awareness among ED staff (option 1) is critical because the ED is often the first point of contact for patients with undiagnosed or active TB, especially those from high-prevalence regions. Increased awareness can improve early identification, isolation, and reporting of potential cases. Reviewing the TB exposure control plan (option 4) is equally important, as it allows the infection preventionist to assess and update protocols—such as ventilation, personal protective equipment (PPE) use, and screening processes—to address the heightened risk posed by the growing number of cases among foreign-born individuals (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.2 - Implement measures to prevent transmission of infectious agents).
Option 2 (inform staff who are foreign-born) is not the best use of this data, as the information pertains to patient demographics rather than staff risk, and targeting staff based on their origin could be inappropriate without specific exposure evidence. Option 3 (educate patients and visitors) is a general education strategy but less directly actionable with this specific epidemiological data, which is more relevant to healthcare worker preparedness and facility protocols. Combining options 1 and 4 aligns with CBIC’s emphasis on using surveillance data to guide prevention and control measures, ensuring a proactive response to the increased TB burden (CBIC Practice Analysis, 2022, Domain II: Surveillance and Epidemiologic Investigation, Competency 2.5 - Use data to guide infection prevention and control strategies).
Therapeutic antimicrobial agents should be used when
Options:
the infecting agent is unknown
the patient's illness warrants treatment prior to culture results
the patient symptoms suggest likely pathogens.
Following identification of the pathogen and sensitives.
Answer:
DExplanation:
Therapeutic antimicrobial agents should ideally be pathogen-directed to minimize resistance, side effects, and treatment failure. Once the causative pathogen and its antimicrobial susceptibilities are known, the most narrow-spectrum, effective agent should be used.
Why the Other Options Are Incorrect?
A. The infecting agent is unknown – Empiric therapy may be necessary initially, but definitive therapy should be based on pathogen identification.
B. The patient's illness warrants treatment prior to culture results – This applies to empiric therapy, but not to definitive antimicrobial selection.
C. The patient’s symptoms suggest likely pathogens – Clinical presentation guides empiric treatment, but definitive therapy should follow culture and susceptibility testing.
CBIC Infection Control Reference
APIC emphasizes the importance of selecting antimicrobials based on pathogen identification and susceptibility testing to prevent antimicrobial resistance.
A hospital is experiencing an increase in multidrug-resistant Acinetobacter baumannii infections in the intensive care unit (ICU). The infection preventionist's FIRST action should be to:
Options:
Implement universal contact precautions for all ICU patients.
Conduct an epidemiologic investigation to identify potential sources.
Perform environmental sampling to detect Acinetobacter on surfaces.
Initiate decolonization protocols for all ICU patients.
Answer:
BExplanation:
Epidemiologic Investigation:
The first step in an outbreak response is to characterize cases by person, place, and time.
Identifying common exposures (e.g., ventilators, catheters, or contaminated surfaces) helps determine the source.
Why Other Options Are Incorrect:
A. Universal contact precautions: Premature; precautions should be tailored based on transmission patterns.
C. Environmental sampling: Should be done after identifying epidemiologic links.
D. Decolonization protocols: Not routinely recommended for Acinetobacter outbreaks.
CBIC Infection Control References:
CIC Study Guide, "Epidemiologic Investigations in Outbreaks," Chapter 4.
When developing an exposure control plan, the MOST important aspect in the prevention of exposure to tuberculosis is:
Options:
Placement of the patient in an airborne infection isolation room.
Identification of a potentially infectious patient.
Prompt initiation of chemotherapeutic agents.
Use of personal protective equipment.
Answer:
BExplanation:
Tuberculosis (TB), caused by Mycobacterium tuberculosis, is an airborne disease that poses a significant risk in healthcare settings, particularly through exposure to infectious droplets. The Certification Board of Infection Control and Epidemiology (CBIC) emphasizes the "Prevention and Control of Infectious Diseases" domain, which includes developing exposure control plans, aligning with the Centers for Disease Control and Prevention (CDC) "Guidelines for Preventing the Transmission of Mycobacterium tuberculosis in Healthcare Settings" (2005). The question seeks the most important aspect of an exposure control plan to prevent TB exposure, requiring a prioritization of preventive strategies.
Option B, "Identification of a potentially infectious patient," is the most important aspect. Early identification of individuals with suspected or confirmed TB (e.g., through symptom screening like persistent cough, fever, or weight loss, or diagnostic tests like chest X-rays and sputum smears) allows for timely isolation and treatment, preventing further transmission. The CDC guidelines stress that the first step in an exposure control plan is to recognize patients with signs or risk factors for infectious TB, as unrecognized cases are the primary source of healthcare worker and patient exposures. The Occupational Safety and Health Administration (OSHA) also mandates risk assessment and early detection as foundational to TB control plans.
Option A, "Placement of the patient in an airborne infection isolation room," is a critical control measure once a potentially infectious patient is identified. Airborne infection isolation rooms (AIIRs) with negative pressure ventilation reduce the spread of infectious droplets, as recommended by the CDC. However, this step depends on prior identification; placing a patient in an AIIR without knowing their infectious status is inefficient and not the initial priority. Option C, "Prompt initiation of chemotherapeutic agents," is essential for treating active TB and reducing infectiousness, typically within days of effective therapy, per CDC guidelines. However, this follows identification and diagnosis (e.g., via acid-fast bacilli smear or culture), making it a secondary action rather than the most important preventive aspect. Option D, "Use of personal protective equipment," such as N95 respirators, is a key protective measure for healthcare workers once an infectious patient is identified, as outlined by the CDC and OSHA. However, PPE is a reactive measure that mitigates exposure after identification and isolation, not the foundational step to prevent it.
The CBIC Practice Analysis (2022) and CDC guidelines prioritize early identification as the cornerstone of TB exposure prevention, enabling all subsequent interventions. Option B ensures that the exposure control plan addresses the source of transmission at its outset, making it the most important aspect.
Which of the following management activities should be performed FIRST?
Options:
Evaluate project results
Establish goals
Plan and organize activities
Assign responsibility for projects
Answer:
BExplanation:
To determine which management activity should be performed first, we need to consider the logical sequence of steps in effective project or program management, particularly in the context of infection control as guided by CBIC principles. Management activities typically follow a structured process, and the order of these steps is critical to ensuring successful outcomes.
A. Evaluate project results: Evaluating project results involves assessing the outcomes and effectiveness of a project after its implementation. This step relies on having completed the project or at least reached a stage where outcomes can be measured. Performing this activity first would be premature, as there would be no results to evaluate without prior planning, goal-setting, and execution. Therefore, this cannot be the first step.
B. Establish goals: Establishing goals is the foundational step in any management process. Goals provide direction, define the purpose, and set the criteria for success. In the context of infection control, as emphasized by CBIC, setting clear objectives (e.g., reducing healthcare-associated infections by a specific percentage) is essential before any other activities can be planned or executed. This step aligns with the initial phase of strategic planning, making it the logical first activity. Without established goals, subsequent steps lack focus and purpose.
C. Plan and organize activities: Planning and organizing activities involve developing a roadmap to achieve the goals, including timelines, resources, and tasks. This step depends on having clear goals to guide the planning process. In infection control, this might include designing interventions to meet infection reduction targets. While critical, it cannot be the first step because planning requires a predefined objective to be effective.
D. Assign responsibility for projects: Assigning responsibility involves delegating tasks and roles to individuals or teams. This step follows the establishment of goals and planning, as responsibilities need to be aligned with the specific objectives and organized activities. In an infection control program, this might mean assigning staff to monitor compliance with hand hygiene protocols. Doing this first would be inefficient without a clear understanding of the goals and plan.
The correct sequence in management, especially in a structured field like infection control, begins with establishing goals to provide a clear target. This is followed by planning and organizing activities, assigning responsibilities, and finally evaluating results. The CBIC framework supports this approach by emphasizing the importance of setting measurable goals as part of the infection prevention and control planning process, which is a prerequisite for all subsequent actions.
CBIC Infection Prevention and Control (IPC) Core Competency Model (updated 2023), Domain V: Management and Communication, which highlights the importance of setting goals as the initial step in managing infection control programs.
CBIC Examination Content Outline, Domain V: Leadership and Program Management, which underscores the need for goal-setting prior to planning and implementation of infection control initiatives.
An employee is presenting to Occupational Health for clearance prior to starting work at a healthcare facility. They have a history of having received the Bacillus Calmette-Guérin (BCG) vaccination. What is the preferred methodology for pre-work clearance?
Options:
Referral to tuberculosis (TB) clinic
Initial chest radiograph
Interferon-gamma release assay
Two-step purified protein derivative-based Tuberculin skin test (TST)
Answer:
CExplanation:
The preferred methodology for pre-work clearance in this scenario is the interferon-gamma release assay (IGRA), making option C the correct choice. This conclusion is supported by the guidelines from the Certification Board of Infection Control and Epidemiology (CBIC), which align with recommendations from the Centers for Disease Control and Prevention (CDC) for tuberculosis (TB) screening in healthcare workers. The employee’s history of receiving the Bacillus Calmette-Guérin (BCG) vaccination, a vaccine commonly used in some countries to prevent severe forms of TB, is significant because it can cause false-positive results in the traditional Tuberculin skin test (TST) due to cross-reactivity with BCG antigens (CBIC Practice Analysis, 2022, Domain I: Identification of Infectious Disease Processes, Competency 1.3 - Apply principles of epidemiology).
The IGRA, such as the QuantiFERON-TB Gold test, measures the release of interferon-gamma from T-cells in response to specific TB antigens (e.g., ESAT-6 and CFP-10) that are not present in BCG or most non-tuberculous mycobacteria. This makes it a more specific and reliable test for detecting latent TB infection (LTBI) in individuals with a history of BCG vaccination, avoiding the false positives associated with the TST. The CDC recommends IGRA over TST for BCG-vaccinated individuals when screening for TB prior to healthcare employment (CDC Guidelines for Preventing Transmission of Mycobacterium tuberculosis, 2005, updated 2019).
Option A (referral to tuberculosis clinic) is a general action but not a specific methodology for clearance; it may follow testing if results indicate further evaluation is needed. Option B (initial chest radiograph) is used to detect active TB disease rather than latent infection and is not a primary screening method for pre-work clearance, though it may be indicated if IGRA results are positive. Option D (two-step purified protein derivative-based Tuberculin skin test) is less preferred because the BCG vaccination can lead to persistent cross-reactivity, reducing its specificity and reliability in this context. The two-step TST is typically used to establish a baseline in unvaccinated individuals with potential prior exposure, but it is not ideal for BCG-vaccinated individuals.
The IP’s role includes ensuring accurate TB screening to protect both the employee and patients, aligning with CBIC’s focus on preventing transmission of infectious diseases in healthcare settings (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.2 - Implement measures to prevent transmission of infectious agents).
What is the limitation of using liquid chemical sterilants to sterilize medical items?
Options:
It does not kill the spores.
The sterility is not maintained during storage.
It requires a contact time of at least 12 hours.
It can only be used for heat tolerant devices.
Answer:
BExplanation:
The correct answer is B, "The sterility is not maintained during storage," as this represents a key limitation of using liquid chemical sterilants to sterilize medical items. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines and standards from the Association for the Advancement of Medical Instrumentation (AAMI), liquid chemical sterilants, such as glutaraldehyde or peracetic acid, are effective for sterilizing heat-sensitive medical devices by eliminating all forms of microbial life, including spores, when used according to manufacturer instructions (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.3 - Ensure safe reprocessing of medical equipment). However, a significant limitation is that sterility is not guaranteed after the items are removed from the sterilant and stored, as the sterile barrier can be compromised by environmental contamination, improper packaging, or handling (AAMI ST58:2013, Chemical Sterilization and High-Level Disinfection in Health Care Facilities).
Option A (it does not kill the spores) is incorrect because liquid chemical sterilants are designed to achieve sterilization, including the destruction of bacterial spores, provided the contact time, concentration, and conditions specified by the manufacturer are met. Option C (it requires a contact time of at least 12 hours) is not a universal limitation; while some liquid sterilants require extended contact times (e.g., 10-12 hours for certain formulations), this is a procedural requirement rather than an inherent limitation, and shorter times may be sufficient with other agents or automated systems. Option D (it can only be used for heat tolerant devices) is incorrect because liquid chemical sterilants are specifically intended for heat-sensitive devices that cannot withstand steam or dry heat sterilization.
The limitation of sterility not being maintained during storage underscores the need for immediate use of sterilized items or the use of proper sterile packaging and storage protocols to prevent recontamination. This aligns with CBIC’s focus on ensuring the safety and efficacy of reprocessed medical equipment in infection prevention (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.3 - Ensure safe reprocessing of medical equipment). Healthcare facilities must implement strict post-sterilization handling and storage practices to mitigate this limitation.
Which of the following statements characterizes the proper use of chemical disinfectants?
Options:
All items to be processed must be cleaned prior to being submerged in solution.
The label on the solution being used must indicate that it kills all viable micro-organisms.
The solution should be adaptable for use as an antiseptic.
A chemical indicator must be used with items undergoing high-level disinfection.
Answer:
AExplanation:
The proper use of chemical disinfectants is a critical aspect of infection control, as outlined by the Certification Board of Infection Control and Epidemiology (CBIC). Chemical disinfectants are used to eliminate or reduce pathogenic microorganisms on inanimate objects, and their effective application requires adherence to specific protocols to ensure safety and efficacy. Let’s evaluate each option based on infection control standards:
A. All items to be processed must be cleaned prior to being submerged in solution.: This statement is a fundamental principle of disinfectant use. Cleaning (e.g., removing organic material such as blood, tissue, or dirt) is a prerequisite before disinfection because organic matter can inactivate or reduce the effectiveness of chemical disinfectants. The CBIC emphasizes that proper cleaning is the first step in the disinfection process to ensure that disinfectants can reach and kill microorganisms. This step is universally required for all levels of disinfection (low, intermediate, and high), making it a characterizing feature of proper use.
B. The label on the solution being used must indicate that it kills all viable micro-organisms.: This statement is misleading. No disinfectant can be guaranteed to kill 100% of all viable microorganisms under all conditions, as efficacy depends on factors like contact time, concentration, and the presence of organic material. Disinfectant labels typically indicate the types of microorganisms (e.g., bacteria, viruses, fungi) and the level of disinfection (e.g., high-level, intermediate-level) they are effective against, based on standardized tests (e.g., EPA or FDA guidelines). Claiming that a solution kills all viable microorganisms is unrealistic and not a requirement for proper use; instead, the label must specify the intended use and efficacy, which varies by product.
C. The solution should be adaptable for use as an antiseptic.: An antiseptic is a chemical agent used on living tissue (e.g., skin) to reduce microbial load, whereas a disinfectant is used on inanimate surfaces. While some chemicals (e.g., alcohol) can serve both purposes, this is not a requirement for proper disinfectant use. The adaptability of a solution for antiseptic use is irrelevant to its classification or application as a disinfectant, which focuses on environmental or equipment decontamination. This statement does not characterize proper disinfectant use.
D. A chemical indicator must be used with items undergoing high-level disinfection.: Chemical indicators (e.g., test strips or tapes) are used to verify that the disinfection process has met certain parameters (e.g., concentration or exposure time), particularly in sterilization or high-level disinfection (HLD). While this is a recommended practice for quality assurance in HLD (e.g., with glutaraldehyde or hydrogen peroxide), it is not a universal requirement for all chemical disinfectant use. HLD applies specifically to semi-critical items (e.g., endoscopes), and the need for indicators depends on the protocol and facility standards. This statement is too narrow and specific to characterize the proper use of chemical disinfectants broadly.
The correct answer is A, as cleaning prior to disinfection is a foundational and universally applicable step in the proper use of chemical disinfectants. This aligns with CBIC guidelines, which stress the importance of a clean surface to maximize disinfectant efficacy and prevent infection transmission in healthcare settings.
CBIC Infection Prevention and Control (IPC) Core Competency Model (updated 2023), Domain IV: Environment of Care, which mandates cleaning as a prerequisite for effective disinfection.
CBIC Examination Content Outline, Domain III: Prevention and Control of Infectious Diseases, which includes protocols for the proper use of disinfectants, emphasizing pre-cleaning.
CDC Guidelines for Disinfection and Sterilization in Healthcare Facilities (2021), which reinforce that cleaning must precede disinfection to ensure efficacy.
Which of the following community-acquired infections has the greatest potential public health impact?
Options:
Cryptosporidium enteritis
Fifth disease (parvovirus B-19)
Clostridial myositis (gas gangrene)
Cryptococcal meningitis
Answer:
AExplanation:
The correct answer is A, "Cryptosporidium enteritis," as it has the greatest potential public health impact among the listed community-acquired infections. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, the public health impact of an infection is determined by factors such as its transmissibility, severity, population at risk, and potential for outbreaks. Cryptosporidium enteritis, caused by the protozoan parasite Cryptosporidium, is a waterborne illness that spreads through contaminated water or food, leading to severe diarrhea, particularly in immunocompromised individuals. Its significant public health impact stems from its high transmissibility in community settings (e.g., via recreational water or daycare centers), the difficulty in eradicating the oocysts with standard chlorination, and the potential to cause large-scale outbreaks affecting vulnerable populations, such as children or the elderly (CBIC Practice Analysis, 2022, Domain I: Identification of Infectious Disease Processes, Competency 1.3 - Apply principles of epidemiology). This is exemplified by notable outbreaks, such as the 1993 Milwaukee outbreak affecting over 400,000 people.
Option B (Fifth disease, caused by parvovirus B-19) is a viral infection primarily affecting children, causing a mild rash and flu-like symptoms. While it can pose risks to pregnant women (e.g., fetal anemia), it is generally self-limiting and has limited community-wide transmission potential, reducing its public health impact. Option C (clostridial myositis, or gas gangrene, caused by Clostridium perfringens) is a severe but rare infection typically associated with traumatic wounds or surgery, with limited person-to-person spread, making its public health impact low due to its sporadic nature. Option D (cryptococcal meningitis, caused by Cryptococcus neoformans) primarily affects immunocompromised individuals (e.g., those with HIV/AIDS) and is not highly transmissible in the general community, confining its impact to specific at-risk groups rather than the broader population.
The selection of Cryptosporidium enteritis aligns with CBIC’s focus on identifying infections with significant epidemiological implications, enabling infection preventionists to prioritize surveillance and control measures for diseases with high outbreak potential (CBIC Practice Analysis, 2022, Domain II: Surveillance and Epidemiologic Investigation, Competency 2.1 - Conduct surveillance for healthcare-associated infections and epidemiologically significant organisms). This is supported by CDC data highlighting waterborne pathogens as major public health concerns (CDC Parasites - Cryptosporidium, 2023).
An infection preventionist receives a call from a healthcare personnel (HCP) who is in the first trimester of pregnancy and has cared for a patient with cytomegalovirus (CMV) pneumonitis. Which of the following actions is MOST appropriate?
Options:
Recommend the pregnant HCP receive immediate post-exposure follow-up.
Reassign the HCP to a low-risk patient care area until second trimester.
Emphasize consistent adherence to Standard Precautions.
Instruct the HCP to wear a mask during patient care activities.
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) states that cytomegalovirus (CMV) is a common virus transmitted through direct contact with body fluids, including saliva, urine, blood, and respiratory secretions. In healthcare settings, Standard Precautions are sufficient to prevent CMV transmission, even for pregnant healthcare personnel.
Importantly, routine reassignment, work restriction, or removal from patient care is not recommended for pregnant HCP caring for patients with CMV, including those in the first trimester. The Study Guide emphasizes that the most effective preventive measure is strict adherence to Standard Precautions, particularly hand hygiene and appropriate use of personal protective equipment when contact with body fluids is anticipated. These measures have been shown to significantly reduce the risk of CMV acquisition.
Option A is incorrect because there is no indication for immediate post-exposure evaluation in the absence of a recognized exposure such as a needlestick or mucous membrane contact. Option B is not supported by evidence or guidelines and may contribute to unnecessary workforce restrictions. Option D is insufficient and misleading, as CMV is not transmitted via the airborne route and masking alone does not address the primary transmission risks.
For CIC® exam preparation, it is critical to recognize that education and reinforcement of Standard Precautions—not work exclusion—are the cornerstone of CMV prevention for pregnant healthcare workers.
Surgical site infection (SSI) data for the previous quarter reveal the following numbers. The surgeon with the highest infection rate is Doctor
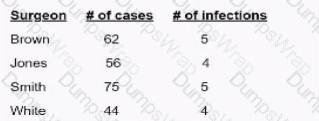
Options:
Brown
Jones.
Smith
White
Answer:
DExplanation:
To determine which surgeon has the highest surgical site infection (SSI) rate, use the following formula:
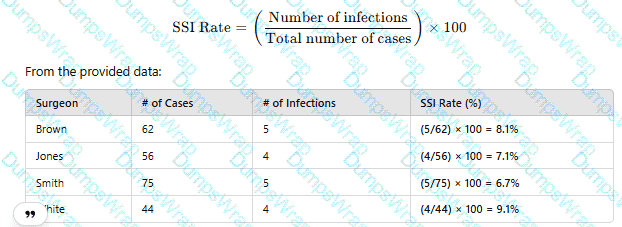
Since Dr. White has the highest SSI rate at 9.1%, the correct answer is D. White.
CBIC Infection Control Reference
SSI rates are calculated using infection count per total procedures and reported as percentage values.
What inflammatory reaction may occur in the eye after cataract surgery due to a breach in disinfection and sterilization of intraocular surgical instruments?
Options:
Endophthalmitis
Bacterial conjunctivitis
Toxic Anterior Segment Syndrome
Toxic Posterior Segment Syndrome
Answer:
CExplanation:
The correct answer is C, "Toxic Anterior Segment Syndrome," as this is the inflammatory reaction that may occur in the eye after cataract surgery due to a breach in disinfection and sterilization of intraocular surgical instruments. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, Toxic Anterior Segment Syndrome (TASS) is a sterile, acute inflammatory reaction that can result from contaminants introduced during intraocular surgery, such as endotoxins, residues from improper cleaning, or chemical agents left on surgical instruments due to inadequate disinfection or sterilization processes (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.3 - Ensure safe reprocessing of medical equipment). TASS typically presents within 12-48 hours post-surgery with symptoms like pain, redness, and anterior chamber inflammation, and it is distinct from infectious causes because it is not microbial in origin. A breach in reprocessing protocols, such as failure to remove detergents or improper sterilization, is a known risk factor, making it highly relevant to infection prevention efforts in surgical settings.
Option A (endophthalmitis) is an infectious inflammation of the internal eye structures, often caused by bacterial or fungal contamination, which can also result from poor sterilization but is distinguished from TASS by its infectious nature and longer onset (days to weeks). Option B (bacterial conjunctivitis) affects the conjunctiva and is typically a surface infection unrelated to intraocular surgery or sterilization breaches of surgical instruments. Option D (toxic posterior segment syndrome) is not a recognized clinical entity in the context of cataract surgery; inflammation in the posterior segment is more commonly associated with infectious endophthalmitis or other conditions, not specifically linked to reprocessing failures.
The focus on TASS aligns with CBIC’s emphasis on ensuring safe reprocessing to prevent adverse outcomes in surgical patients, highlighting the need for rigorous infection control measures (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.5 - Evaluate the environment for infection risks). This is supported by CDC and American Academy of Ophthalmology guidelines, which identify TASS as a preventable complication linked to reprocessing errors (CDC Guidelines for Disinfection and Sterilization, 2019; AAO TASS Task Force Report, 2017).
When assessing a patient’s infection prevention and control educational needs, it is necessary to FIRST determine the patient’s
Options:
severity of illness.
educational background.
duration of hospitalization.
baseline knowledge of the subject.
Answer:
DExplanation:
The correct answer is D, "baseline knowledge of the subject," as this is the necessary first step when assessing a patient’s infection prevention and control educational needs. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, effective patient education in infection prevention and control requires a tailored approach that begins with understanding the patient’s existing knowledge and comprehension of the topic. Determining baseline knowledge allows the infection preventionist (IP) to identify gaps, customize educational content to the patient’s level of understanding, and ensure the information is relevant and actionable (CBIC Practice Analysis, 2022, Domain IV: Education and Research, Competency 4.1 - Develop and implement educational programs). This step ensures that education is neither too basic nor overly complex, maximizing its effectiveness in promoting behaviors such as hand hygiene, wound care, or adherence to isolation protocols.
Option A (severity of illness) is an important clinical consideration that may influence the timing or method of education delivery, but it is not the first step in assessing educational needs. The severity might affect the patient’s ability to learn, but it does not directly inform the content or starting point of the education. Option B (educational background) provides context about the patient’s general learning capacity (e.g., literacy level or language preference), but it is secondary to assessing specific knowledge about infection prevention, as background alone does not reveal current understanding. Option C (duration of hospitalization) may impact the opportunity for education but is not a primary factor in determining what the patient needs to learn; it is more relevant to scheduling or prioritizing educational interventions.
The focus on baseline knowledge aligns with adult learning principles endorsed by CBIC, which emphasize assessing learners’ prior knowledge to build effective educational strategies (CBIC Practice Analysis, 2022, Domain IV: Education and Research, Competency 4.2 - Evaluate the effectiveness of educational programs). This approach ensures patient-centered care and supports infection control by empowering patients with the knowledge to participate in their own prevention efforts.
An outbreak of Candida auris is suspected in the infection preventionist's (IP) facility. The IP's investigation must be conducted in a standard method and communication is critical. Which first step is MOST important?
Options:
Conduct environmental cultures
Plan to prevent future outbreaks
Notify facility administration
Perform analytical studies
Answer:
CExplanation:
In an outbreak investigation, the first critical step is to notify facility administration and other key stakeholders. This ensures the rapid mobilization of resources, coordination with infection control teams, and compliance with regulatory reporting requirements.
Why the Other Options Are Incorrect?
A. Conduct environmental cultures – While environmental sampling may be necessary, it is not the first step. The outbreak must first be confirmed and administration alerted.
B. Plan to prevent future outbreaks – Prevention planning happens later after the outbreak has been investigated and controlled.
D. Perform analytical studies – Data analysis occurs after case definition and initial response measures are in place.
CBIC Infection Control Reference
APIC guidelines state that the first step in an outbreak investigation is confirming the outbreak and notifying key stakeholders.
Which of the following BEST reduces the risk of infection in patients undergoing peritoneal dialysis?
Options:
Daily dressing change of the catheter exit site
Weekly surveillance cultures of catheter exit sites
Catheter insertion in the operating room
Irrigation of catheter with antimicrobials between dialysis treatments
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that the most effective strategy for reducing infection risk in peritoneal dialysis (PD) patients is ensuring optimal conditions at the time of catheter insertion. Placement of the peritoneal dialysis catheter in the operating room provides a controlled, sterile environment that minimizes microbial contamination and significantly reduces the risk of early peritonitis and exit-site infections.
Peritoneal dialysis–associated infections are most often linked to contamination during catheter insertion or manipulation. Performing catheter insertion in the operating room allows for strict adherence to aseptic technique, appropriate airflow controls, surgical hand antisepsis, and use of sterile instruments—all of which are essential infection prevention measures highlighted in the Study Guide.
The other options are less effective or not recommended. Daily dressing changes (Option A) may actually increase manipulation of the exit site and raise infection risk if not clinically indicated. Weekly surveillance cultures (Option B) are not recommended, as they do not prevent infection and may lead to unnecessary antimicrobial use. Irrigating catheters with antimicrobials (Option D) is discouraged because it has not been shown to reduce infection rates and may contribute to antimicrobial resistance.
For the CIC® exam, it is important to recognize that prevention of peritoneal dialysis–associated infection begins with proper catheter placement under optimal sterile conditions, making operating room insertion the best answer.
==========
An infection preventionist is asked by the Central Supply department to review its process for assigning expiration dates to sterile supplies. Which of the following is the MOST important consideration?
Options:
Sterility is related to package integrity.
Items must have 30- to 90-day expiration dates.
The expiration date depends on the type of packaging.
The expiration date depends on the type of sterilization.
Answer:
AExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that the most important consideration in assigning expiration dates to sterile supplies is package integrity, reflecting the principle of event-related sterility. Modern infection prevention practice recognizes that sterility is not determined by time alone but by whether an event has occurred that compromises the sterile barrier system.
Sterile items remain sterile indefinitely as long as the packaging remains intact, dry, and properly stored, and no contamination event (such as tearing, puncture, moisture exposure, or improper handling) has occurred. Therefore, the presence or absence of a printed expiration date is less important than assurance that the package integrity has been maintained throughout storage and handling.
Option A is correct because it captures the foundational concept that sterility is directly linked to the integrity of the packaging, not an arbitrary time frame. Option B is incorrect because fixed time-based expiration dates (e.g., 30–90 days) are outdated and not evidence-based. Options C and D may influence packaging durability or compatibility with sterilization methods, but they are secondary considerations and do not override the primary determinant of sterility.
For the CIC® exam, this question reinforces a critical sterilization principle: event-related shelf life is the standard, and infection preventionists should focus on policies that emphasize package integrity, proper storage conditions, and handling practices rather than routine time-based expiration dating.
The infection preventionist notes an increase in Clostridioides difficile infections (CDI) in the ICU. A Root Cause Analysis (RCA) is scheduled. What is the goal of a Root Cause Analysis?
Options:
Proactively identify potential failures.
Identify processes to prevent recurrence.
Determine strengths, weaknesses, opportunities, and threats.
Educate staff in order to avoid individual blame.
Answer:
BExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) defines a Root Cause Analysis (RCA) as a retrospective, systematic process used to understand why an adverse event or undesired outcome occurred and what system-level changes are needed to prevent it from happening again. In the context of an increase in Clostridioides difficile infections in an ICU, the primary goal of an RCA is to identify underlying process failures and implement corrective actions to prevent recurrence.
RCA focuses on systems and processes rather than individual performance. Through structured methods such as event mapping, cause-and-effect analysis, and contributing factor review, the team examines elements such as antimicrobial use, environmental cleaning practices, hand hygiene compliance, isolation implementation, diagnostic testing practices, and workflow design. The ultimate outcome of an RCA is a set of actionable, sustainable process improvements that reduce the likelihood of similar events in the future.
Option A describes Failure Mode and Effects Analysis (FMEA), which is a proactive risk assessment tool. Option C refers to a SWOT analysis, used for strategic planning rather than event investigation. Option D reflects an important principle of RCA culture (non-punitive), but it is not the primary goal.
For the CIC® exam, it is essential to recognize that the core purpose of RCA is preventing recurrence through system improvement, making option B the correct answer.
==========
A facility performs active surveillance cultures for methicillin-resistant Staphylococcus aureus (MRSA) on all patients upon admission and weekly. Twenty-two of the patients with positive cultures for MRSA were transferred from outlying facilities. The following MRSA data for a 3-month period are shown. Review of the data reveals which of the following is increasing?

Options:
Transmission of MRSA on the unit
Prevalence of MRSA at admission
Patients with MRSA infection
Compliance with collecting MRSA cultures
Answer:
BExplanation:
The table separates admission cultures from weekly cultures, which is a common surveillance approach to distinguish imported MRSA burden (present on admission) from healthcare acquisition (newly detected later). The admission culture percent positive rises over the three months: 14% (Feb) → 18% (Mar) → 19% (Apr). That pattern indicates an increasing admission prevalence (option B). NHSN MDRO surveillance methods describe admission prevalence as a proxy measure using admission-related data to quantify organisms present at the time of entry into a location/facility.
By contrast, weekly culture positivity—often used as a proxy for on-unit acquisition/transmission when admission screening is in place—decreases: 6% → 5.6% → 4%, so option A is not increasing. The dataset also does not provide information about MRSA infections versus colonization (so C cannot be concluded), nor does it provide a denominator for “compliance” (e.g., expected admissions/weekly screens completed), so D cannot be determined. This interpretation aligns with standard infection prevention use of MRSA surveillance data to track prevalence (burden) versus incidence/acquisition.
Each item or package that is prepared for sterilization should be labeled with the
Options:
storage location.
type of sterilization process.
sterilizer identification number or code.
cleaning method (e.g., mechanical or manual).
Answer:
CExplanation:
The correct answer is C, "sterilizer identification number or code," as this is the essential information that each item or package prepared for sterilization should be labeled with. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, proper labeling of sterilized items is a critical component of infection prevention and control to ensure traceability and verify the sterilization process. The sterilizer identification number or code links the item to a specific sterilization cycle, allowing the infection preventionist (IP) and sterile processing staff to track the equipment used, confirm compliance with standards (e.g., AAMI ST79), and facilitate recall or investigation if issues arise (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.3 - Ensure safe reprocessing of medical equipment). This labeling ensures that the sterility of the item can be assured and documented, protecting patient safety by preventing the use of inadequately processed items.
Option A (storage location) is important for inventory management but is not directly related to the sterilization process itself and does not provide evidence of the sterilization event. Option B (type of sterilization process) indicates the method (e.g., steam, ethylene oxide), which is useful but less critical than the sterilizer identification, as the process type alone does not confirm the specific cycle or equipment used. Option D (cleaning method, e.g., mechanical or manual) is a preliminary step in reprocessing, but it is not required on the sterilization label, as the focus shifts to sterilization verification once the item is prepared.
The requirement for a sterilizer identification number or code aligns with CBIC’s emphasis on maintaining rigorous tracking and quality assurance in the reprocessing of medical devices, ensuring accountability and adherence to best practices (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.5 - Evaluate the environment for infection risks). This practice is mandated by standards such as AAMI ST79 to support effective infection control in healthcare settings.
Which of the following is an example of a syndromic surveillance indicator?
Options:
Number of individuals presenting with influenza-like illness in the emergency department each day
Number of individuals presenting with laboratory-confirmed influenza in the emergency department each day
Rate of central line–associated bloodstream infections each quarter
Number of cases of methicillin-resistant Staphylococcus aureus in an intensive care unit each month
Answer:
AExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) describes syndromic surveillance as a surveillance method that focuses on clinical signs, symptoms, or health-seeking behaviors rather than confirmed diagnoses. Its primary purpose is early detection of outbreaks or emerging health threats, often before laboratory confirmation is available.
Option A is the correct example because tracking the number of individuals presenting with influenza-like illness (ILI) relies on symptom patterns such as fever, cough, and sore throat. These data are typically collected in near real time from emergency department chief complaints or triage notes, allowing infection preventionists and public health authorities to identify unusual increases quickly and initiate early response measures.
Option B is not syndromic surveillance because it depends on laboratory-confirmed diagnoses, which are characteristic of traditional, diagnosis-based surveillance. Option C represents device-associated infection surveillance, which is retrospective and outcome-focused. Option D involves laboratory-confirmed antimicrobial-resistant organisms and is also not syndromic.
For CIC® exam preparation, it is important to remember that syndromic surveillance prioritizes speed over diagnostic certainty. By monitoring symptom clusters rather than confirmed cases, it enables earlier recognition of outbreaks such as influenza, gastrointestinal illness, or bioterrorism-related events, making it a critical component of public health preparedness and response.
During the past week, three out of four blood cultures from a febrile neonate in an intensive care unit grew coagulase-negative staphylococci. This MOST likely indicates:
Options:
Laboratory error.
Contamination.
Colonization.
Infection.
Answer:
BExplanation:
The scenario involves a febrile neonate in an intensive care unit (ICU) with three out of four blood cultures growing coagulase-negative staphylococci (CoNS) over the past week. The Certification Board of Infection Control and Epidemiology (CBIC) emphasizes accurate interpretation of microbiological data in the "Identification of Infectious Disease Processes" domain, aligning with the Centers for Disease Control and Prevention (CDC) guidelines for healthcare-associated infections. Determining whether this represents a true infection, contamination, colonization, or laboratory error requires evaluating the clinical and microbiological context.
Option B, "Contamination," is the most likely indication. Coagulase-negative staphylococci, such as Staphylococcus epidermidis, are common skin flora and frequent contaminants in blood cultures, especially in neonates where skin preparation or sampling technique may be challenging. The CDC’s "Guidelines for the Prevention of Intravascular Catheter-Related Infections" (2017) and the Clinical and Laboratory Standards Institute (CLSI) note that multiple positive cultures (e.g., two or more) are typically required to confirm true bacteremia, particularly with CoNS, unless accompanied by clear clinical signs of infection (e.g., worsening fever, hemodynamic instability) and no other explanation. The inconsistency (three out of four cultures) and the neonate’s ICU setting—where contamination from skin or catheter hubs is common—suggest that the positive cultures likely result from contamination during blood draw rather than true infection. Studies, such as those in the Journal of Clinical Microbiology (e.g., Beekmann et al., 2005), indicate that CoNS in blood cultures is contaminated in 70-80% of cases when not supported by robust clinical correlation.
Option A, "Laboratory error," is possible but less likely as the primary explanation. Laboratory errors (e.g., mislabeling or processing mistakes) could occur, but the repeated growth in three of four cultures suggests a consistent finding rather than a random error, making contamination a more plausible cause. Option C, "Colonization," refers to the presence of microorganisms on or in the body without invasion or immune response. While CoNS can colonize the skin or catheter sites, colonization does not typically result in positive blood cultures unless there is an invasive process, which is not supported by the data here. Option D, "Infection," is the least likely without additional evidence. True CoNS bloodstream infections (e.g., catheter-related) in neonates are serious but require consistent positive cultures, clinical deterioration (e.g., persistent fever, leukocytosis), and often imaging or catheter removal confirmation. The febrile state alone, with inconsistent culture results, does not meet the CDC’s criteria for diagnosing infection (e.g., at least two positive cultures from separate draws).
The CBIC Practice Analysis (2022) and CDC guidelines stress differentiating contamination from infection to avoid unnecessary treatment, which can drive antibiotic resistance. Given the high likelihood of contamination with CoNS in this context, Option B is the most accurate answer.
A 36-year-old female presents to the Emergency Department with a petechial rash, meningitis, and cardiac arrest. During the resuscitation, a phlebotomist sustained a needlestick injury. The next day, blood cultures reveal Neisseria meningitidis. The exposure management for the phlebotomist is:
Options:
Prophylactic rifampin plus isoniazid.
A tuberculin skin test now and in ten weeks.
Work furlough from day ten to day 21 after exposure.
A review of the phlebotomist’s hepatitis B vaccine status.
Answer:
CExplanation:
The scenario involves a needlestick injury sustained by a phlebotomist during the resuscitation of a patient diagnosed with Neisseria meningitidis infection, characterized by a petechial rash, meningitis, and cardiac arrest. Neisseria meningitidis is a gram-negative diplococcus that can cause meningococcal disease, including meningitis and septicemia, and is transmitted through direct contact with respiratory secretions or, in rare cases, blood exposure. The exposure management for the phlebotomist must align with infection control guidelines, such as those from the Certification Board of Infection Control and Epidemiology (CBIC) and the CDC, to prevent potential infection. Let’s evaluate each option:
A. Prophylactic rifampin plus isoniazid: Prophylactic antibiotics are recommended for close contacts of individuals with meningococcal disease to prevent secondary cases. Rifampin is a standard prophylactic agent for Neisseria meningitidis exposure, typically administered as a 2-day course (e.g., 600 mg every 12 hours for adults). Isoniazid, however, is used for tuberculosis (TB) prophylaxis and is not indicated for meningococcal disease. Combining rifampin with isoniazid is incorrect, as it reflects a confusion with TB management rather than meningococcal exposure. This option is not appropriate.
B. A tuberculin skin test now and in ten weeks: A tuberculin skin test (TST) or interferon-gamma release assay (IGRA) is used to screen for latent tuberculosis infection, with a follow-up test at 8-10 weeks to detect conversion after potential TB exposure. Neisseria meningitidis is not related to TB, and a needlestick injury from a meningococcal patient does not warrant TB testing. This option is irrelevant to the scenario and not the correct exposure management.
C. Work furlough from day ten to day 21 after exposure: Neisseria meningitidis has an incubation period of 2-10 days, with a maximum of about 14 days in rare cases. The CDC and WHO recommend that healthcare workers exposed to meningococcal disease via needlestick or mucosal exposure be monitored for signs of infection (e.g., fever, rash) and, if symptomatic, isolated and treated. Additionally, a work restriction or furlough from day 10 to day 21 after exposure is advised to cover the potential incubation period, especially if prophylaxis is declined or contraindicated. This allows time to observe for symptoms and prevents transmission to vulnerable patients. This is a standard infection control measure and the most appropriate initial management step pending prophylaxis decision.
D. A review of the phlebotomist’s hepatitis B vaccine status: Reviewing hepatitis B vaccine status is a critical step following a needlestick injury, as hepatitis B can be transmitted through blood exposure. However, this applies to bloodborne pathogens (e.g., HBV, HCV, HIV) and is not specific to Neisseria meningitidis, which is primarily a respiratory or mucosal pathogen. While hepatitis B management (e.g., post-exposure prophylaxis with hepatitis B immunoglobulin or vaccine booster) should be addressed as part of a comprehensive needlestick protocol, it is not the first or most relevant priority for meningococcal exposure.
The best answer is C, as the work furlough from day 10 to day 21 after exposure addresses the specific risk of meningococcal disease following a needlestick injury. This aligns with CBIC’s focus on timely intervention and work restriction to prevent transmission in healthcare settings. Prophylactic antibiotics (e.g., rifampin) should also be considered, but the question asks for the exposure management, and furlough is a primary control measure. Hepatitis B and TB considerations are secondary and managed separately.
CBIC Infection Prevention and Control (IPC) Core Competency Model (updated 2023), Domain III: Prevention and Control of Infectious Diseases, which includes protocols for managing exposure to communicable diseases like meningococcal infection.
CBIC Examination Content Outline, Domain IV: Environment of Care, which addresses work restrictions and exposure management.
CDC Guidelines for Meningococcal Disease Prevention and Control (2023), which recommend work furlough and monitoring for exposed healthcare workers.
Passive immunity results from the use of:
Options:
Tetanus antitoxin
Hepatitis B vaccine
Influenza vaccine
Human diploid cell rabies vaccine
Answer:
AExplanation:
The Certification Study Guide (6th edition) defines passive immunity as protection that results from the administration of preformed antibodies, rather than stimulation of the individual’s own immune system. Passive immunity provides immediate but temporary protection, because the recipient does not produce antibodies and therefore does not develop immunologic memory.
Tetanus antitoxin is a classic example of passive immunity. It contains antibodies that neutralize tetanus toxin directly and is used in situations where immediate protection is needed, such as after certain wounds in individuals with unknown or inadequate vaccination history. The study guide emphasizes that passive immunization is particularly important in post-exposure management when waiting for an active immune response would be too slow to prevent disease.
The other options represent active immunization, not passive immunity. Vaccines such as hepatitis B vaccine, influenza vaccine, and human diploid cell rabies vaccine stimulate the recipient’s immune system to produce its own antibodies and immune memory. While rabies immune globulin provides passive immunity, the rabies vaccine itself is an active immunizing agent.
This distinction between active and passive immunity is a frequently tested CIC exam concept, especially in the context of occupational health, post-exposure prophylaxis, and immunization programs. Recognizing that passive immunity involves antibody products (antitoxins or immune globulins) rather than vaccines is essential for accurate infection prevention decision-making.
Which of the following descriptions accurately describes a single-use medical device?
Options:
A device which can be used on a single patient
A device that is sterilized and can be used again on the same patient
A device used on a patient and reprocessed prior to being used again
A device used one time on a patient during a procedure and then discarded
Answer:
DExplanation:
The correct answer is D, "A device used one time on a patient during a procedure and then discarded," as this accurately describes a single-use medical device. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, a single-use device (SUD), also known as a disposable device, is labeled by the manufacturer for one-time use on a patient and is intended to be discarded afterward to prevent cross-contamination and ensure patient safety. This definition is consistent with regulations from the Food and Drug Administration (FDA), which designate SUDs as devices that should not be reprocessed or reused due to risks of infection, material degradation, or failure to restore sterility (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.3 - Ensure safe reprocessing of medical equipment). Examples include certain syringes, catheters, and gloves, which are designed for single use to eliminate the risk of healthcare-associated infections (HAIs).
Option A (a device which can be used on a single patient) is too vague and could apply to both single-use and reusable devices, as reusable devices are also often used on a single patient per procedure before reprocessing. Option B (a device that is sterilized and can be used again on the same patient) describes a reusable device, not a single-use device, as sterilization and reuse are not permitted for SUDs. Option C (a device used on a patient and reprocessed prior to being used again) refers to a reusable device that undergoes reprocessing (e.g., sterilization), which is explicitly prohibited for SUDs under manufacturer and regulatory guidelines.
The focus on discarding after one use aligns with CBIC’s emphasis on preventing infection through adherence to device labeling and safe reprocessing practices, ensuring that healthcare facilities avoid the risks associated with improper reuse of SUDs (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.5 - Evaluate the environment for infection risks). This practice is critical to maintaining a sterile and safe healthcare environment.
Which of the following study designs provides the STRONGEST evidence of a causal relationship between a risk factor and the outcome of interest?
Options:
Randomized clinical trial
Case-control study
Cohort study
Cross-sectional study
Answer:
AExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies the randomized clinical trial (RCT) as the study design that provides the strongest evidence of a causal relationship between a risk factor (or intervention) and an outcome. RCTs are considered the gold standard because they use random assignment to allocate participants to either an intervention group or a control group, which minimizes bias and balances both known and unknown confounding variables between groups.
By controlling exposure and randomly assigning participants, RCTs establish temporality, ensuring that the exposure precedes the outcome—an essential criterion for causality. This design also allows for direct comparison of outcomes under controlled conditions, making it possible to attribute observed differences in outcomes to the intervention or risk factor with a high degree of confidence.
In contrast, cohort studies and case-control studies are observational and can identify associations but are more susceptible to confounding and bias. While cohort studies can demonstrate temporal relationships and estimate risk, they cannot control exposures as precisely as RCTs. Case-control studies are particularly vulnerable to recall and selection bias. Cross-sectional studies assess exposure and outcome simultaneously and cannot establish causation.
For the CIC® exam, it is critical to recognize that randomized clinical trials offer the highest level of evidence for causality, particularly when evaluating interventions, preventive measures, or treatment effectiveness in infection prevention and healthcare epidemiology.
Which of the following options describes a best practice for avoiding occupational health hazards in a healthcare facility?
Options:
The facility is required to conduct annual tuberculosis (TB) testing for healthcare personnel.
The facility is required to conduct varicella post-vaccination testing for healthcare personnel.
The facility should exclude asymptomatic Bordetella pertussis–exposed healthcare personnel from duty.
The facility should provide Neisseria meningitidis pre-exposure vaccination to laboratory personnel.
Answer:
DExplanation:
The Certification Study Guide (6th edition) emphasizes that occupational health hazard prevention is based on risk assessment and targeted protection strategies, particularly for personnel with predictable, high-risk exposures. Providing pre-exposure vaccination against Neisseria meningitidis to laboratory personnel is a recognized best practice because laboratorians who routinely handle N. meningitidis isolates are at increased risk for aerosol or droplet exposure, which can result in rapidly progressive and potentially fatal disease.
The study guide highlights that pre-exposure immunization is preferred over post-exposure management when exposure risk is ongoing and well defined. This strategy aligns with evidence-based occupational health principles and recommendations from public health authorities, making it a proactive and preventive measure rather than a reactive one.
The other options are incorrect because they either reflect outdated practices or inappropriate control measures. Routine annual TB testing is no longer universally required and should be based on facility risk assessment. Post-vaccination varicella serologic testing is not recommended because commercial assays may not reliably detect vaccine-induced immunity. Excluding asymptomatic pertussis-exposed healthcare personnel from duty is not routinely recommended if appropriate prophylaxis is provided.
This question reflects a common CIC exam theme: best practices focus on targeted, evidence-based prevention, especially vaccination strategies for high-risk occupational groups.
The infection preventionist observed a caregiver entering a room without performing hand hygiene. The BEST response would be to
Options:
post additional signage to remind caregivers to wash before entry.
provide immediate feedback and education to the caregiver.
install hand hygiene dispensers in more convenient areas.
design a unit-based education program.
Answer:
BExplanation:
Immediate feedback is a best practice in behavior correction and performance improvement. In hand hygiene non-compliance, real-time intervention allows for immediate correction, education, and reinforcement of infection prevention policies.
The APIC/JCR Workbook recommends:
“Provide simulation training… that provides immediate feedback—for example, how to properly insert a urinary catheter or perform hand hygiene.” This supports behavior change and staff learning.
The APIC Text emphasizes that real-time, direct feedback is more effective than passive measures like signage or delayed education campaigns.
An infection preventionist (IP) is asked to participate on a team to decrease ventilator-associated pneumonia (VAP) rates in a 20-bed ICU. The IP provides the following information. What is the first quarter ventilator utilization ratio?
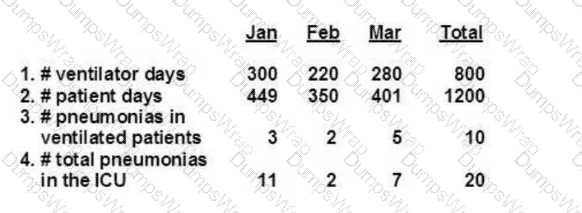
Data Provided (First Quarter):
Ventilator days (Jan–Mar total): 800
Patient days (Jan–Mar total): 1200
Options:
0.13
0.67
1.50
1.67
Answer:
BExplanation:
The Certification Study Guide (6th edition) defines the ventilator utilization ratio (VUR) as a device utilization measure used in surveillance to describe the proportion of patient time during which a specific medical device—in this case, mechanical ventilation—is in use. It is calculated by dividing the total number of ventilator days by the total number of patient days for the same location and time period.
Using the first-quarter data provided, the calculation is as follows:
Ventilator Utilization Ratio = Ventilator Days ÷ Patient Days
Ventilator Utilization Ratio = 800 ÷ 1200 = 0.67
This means that ventilators were in use for 67% of all patient days in the ICU during the first quarter. The study guide emphasizes that device utilization ratios are essential for interpreting device-associated infection data, such as VAP rates, because they reflect the level of patient exposure to the device. Higher utilization increases the population at risk and can influence infection rates independently of prevention practices.
The other answer options are incorrect because they do not reflect the correct calculation. A ratio greater than 1.0 (options C and D) would imply more device days than patient days, which is not possible in this context. Option A underestimates utilization and does not match the provided data.
Understanding and correctly calculating utilization ratios is a core CIC exam competency, as these metrics support accurate surveillance, benchmarking, and performance improvement efforts.
An infection preventionist plans to evaluate 12 months of prospectively collected intensive care unit (ICU) surveillance data on ventilator-associated events. The MOST effective visual representation of these data is a:
Options:
Pareto chart
Control chart
Pie chart
Scatter gram
Answer:
BExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that surveillance data collected over time are best evaluated using statistical process control methods. A control chart is the most effective visual tool for analyzing 12 months of prospectively collected ICU surveillance data on ventilator-associated events (VAEs) because it displays data sequentially over time and distinguishes between normal process variation and significant changes that may require intervention.
Control charts allow infection preventionists to identify trends, shifts, or special cause variation by plotting event rates against calculated control limits. This enables timely recognition of sustained increases or decreases in VAEs and supports data-driven decision-making. Control charts are especially valuable for ongoing surveillance and performance improvement because they demonstrate whether prevention efforts are having a measurable impact.
The other options are less appropriate for this purpose. A Pareto chart is used to prioritize causes contributing to a problem, not to track rates over time. A pie chart shows proportional distribution at a single point in time and does not reflect trends. A scatter gram is used to assess relationships between two variables rather than monitor process stability.
For CIC® exam preparation, it is critical to recognize that when evaluating infection surveillance data longitudinally—particularly for healthcare-associated events—control charts are the preferred and most effective visualization method, aligning with epidemiologic principles and quality improvement methodology outlined in the Study Guide.
==========
Which of the following organisms is associated with a waterborne disease?
Options:
Bacillus anthracis
Cytomegalovirus
Nontuberculous mycobacteria
Stachybotrys
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies nontuberculous mycobacteria (NTM) as organisms commonly associated with waterborne exposure. NTM are environmental mycobacteria widely found in natural and treated water sources, including potable water systems, ice machines, showerheads, faucets, and medical equipment rinsed with tap water. Because these organisms are resistant to standard water disinfection methods and can form biofilms, they are particularly well adapted to survive in plumbing systems.
NTM have been implicated in healthcare-associated infections, especially among immunocompromised patients, and may cause pulmonary disease, skin and soft tissue infections, and invasive disease following exposure to contaminated water or medical devices. The Study Guide emphasizes the importance of water management programs and routine surveillance to prevent waterborne transmission of opportunistic pathogens such as NTM and Legionella.
The other answer options are incorrect. Bacillus anthracis is primarily associated with zoonotic and bioterrorism-related exposure, not waterborne transmission. Cytomegalovirus is transmitted through direct contact with bodily fluids rather than water. Stachybotrys is a mold associated with damp indoor environments but is not considered a waterborne pathogen in the context of infection transmission.
Understanding organisms linked to water systems is critical for infection preventionists, as waterborne pathogens present ongoing risks in healthcare facilities and are a key topic on the CIC® exam.
==========
A patient is Hepatitis B surface antigen (HBsAg) negative, Hepatitis B surface antibody (anti-HBs) positive, and Hepatitis B core antibody (anti-HBc) negative. Which of the following explains these results?
Options:
Response to hepatitis B vaccine series
A recent blood transfusion
Previous hepatitis B infection
Low-level hepatitis B infectiousness
Answer:
AExplanation:
The Certification Study Guide (6th edition) explains that interpretation of hepatitis B serologic markers is a fundamental competency for infection preventionists, particularly in occupational health and exposure management. In this scenario, the patient is HBsAg negative, indicating no current hepatitis B infection; anti-HBs positive, indicating immunity; and anti-HBc negative, meaning there has been no prior natural infection with hepatitis B virus.
This specific serologic pattern is diagnostic of immunity due to vaccination. The hepatitis B vaccine contains only purified hepatitis B surface antigen, not core antigen. As a result, vaccinated individuals develop antibodies to the surface antigen (anti-HBs) but do not develop antibodies to the core antigen (anti-HBc). The study guide emphasizes this distinction as the key factor in differentiating vaccine-induced immunity from immunity due to past infection.
The incorrect options reflect different serologic patterns. Previous hepatitis B infection would produce a positive anti-HBc result. A recent blood transfusion does not confer long-term immunity or this marker pattern. Low-level infectivity would require detectable surface antigen or core antibody.
This question reflects a classic CIC exam topic: recognizing the serologic profile of vaccine-induced immunity. Correct interpretation supports appropriate employee health decisions, post-exposure management, and immunization program evaluation.
Which of the following is the BEST aid in the identification of patients affected by a recall due to failures in endoscope reprocessing?
Options:
Maintaining a log of endoscope use by date of procedure
Maintaining a log of patient identifiers linked with endoscope used
Searching electronic records for endoscope serial number recorded in patient records
Searching electronic records using diagnostic coding to identify all patients that had endoscopy procedures
Answer:
BExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes the importance of traceability in endoscope reprocessing programs to ensure rapid and accurate patient notification when reprocessing failures or recalls occur. The most effective method for identifying affected patients is maintaining a log that directly links each endoscope to specific patient identifiers for every procedure.
This type of tracking system allows infection preventionists to quickly determine exactly which patients were exposed to a particular endoscope during the time period of concern. When reprocessing failures are identified—such as incomplete cleaning, high-level disinfection errors, or equipment malfunction—precise linkage between the endoscope and the patient is essential to limit the scope of exposure investigations, reduce unnecessary notifications, and ensure timely follow-up care.
Option A is insufficient because a date-only log does not identify individual patients. Option C may be useful if serial numbers are consistently documented in the medical record, but this practice is not reliably implemented in many facilities and is therefore less dependable. Option D is overly broad and would identify all patients who underwent endoscopy, rather than those exposed to a specific device, leading to unnecessary alarm and inefficient investigations.
For CIC® exam purposes, understanding that patient-to-device linkage logs are the cornerstone of effective exposure investigation and recall management in endoscope reprocessing is critical and aligns with best-practice infection prevention standards.
During the last week in June, an emergency department log reveals numerous cases of profuse watery diarrhea in individuals 74 years of age and older. During the same time period, four immunocompromised patients were admitted with possible Cryptosporidium. Which of the following actions should the infection preventionist take FIKST?
Options:
Characterize the outbreak by person, place, and time
Increase surveillance facility wide for additional cases
Contact the laboratory to confirm stool identification results
Form a tentative hypothesis about the potential reservoir for this outbreak
Answer:
AExplanation:
When an outbreak of infectious disease is suspected, the first step is to conduct an epidemiologic investigation. This begins with characterizing the outbreak by person, place, and time to establish patterns and trends. This approach, known as descriptive epidemiology, provides critical insights into potential sources and transmission patterns.
Step-by-Step Justification:
Identify Cases and Patterns:
The infection preventionist should analyze patient demographics (person), locations of cases (place), and onset of symptoms (time). This helps in defining the outbreak scope and potential exposure sources.
Create an Epidemic Curve:
An epidemic curve helps determine whether the outbreak is a point-source or propagated event. This can indicate whether the infection is spreading person-to-person or originating from a common source.
Compare with Baseline Data:
Reviewing historical data ensures that the observed cases exceed the expected norm, confirming an outbreak.
Guide Further Investigation:
Establishing basic epidemiologic patterns guides subsequent actions, such as laboratory testing, environmental sampling, and surveillance.
Why Other Options Are Incorrect:
B. Increase surveillance facility-wide for additional cases:
While enhanced surveillance is important, it should follow the initial characterization of the outbreak. Surveillance without a defined case profile may lead to misclassification and misinterpretation.
C. Contact the laboratory to confirm stool identification results:
Confirming lab results is essential but comes after defining the outbreak's characteristics. Without an epidemiologic link, testing may yield results that are difficult to interpret.
D. Form a tentative hypothesis about the potential reservoir for this outbreak:
Hypothesis generation occurs after sufficient epidemiologic data have been collected. Jumping to conclusions without characterization may result in incorrect assumptions and ineffective control measures.
CBIC Infection Control References:
APIC Text, "Outbreak Investigations," Epidemiology, Surveillance, Performance, and Patient Safety Measures.
APIC/JCR Infection Prevention and Control Workbook, Chapter 4, Surveillance Program.
APIC Text, "Investigating Infectious Disease Outbreaks," Guidelines for Epidemic Curve Analysis.
An infection preventionist is reviewing employee health immunization policies. What is the recommendation for tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis (Tdap) for a 55-year-old nurse who received all childhood vaccinations?
Options:
One dose of Tdap vaccine
Two doses of Tdap vaccine at least 14 days apart
Two doses of Tdap vaccine at least 28 days apart
No additional vaccination is recommended
Answer:
AExplanation:
The correct answer is A, "One dose of Tdap vaccine," as this is the recommended immunization for a 55-year-old nurse who received all childhood vaccinations. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, which align with recommendations from the Centers for Disease Control and Prevention (CDC) and the Advisory Committee on Immunization Practices (ACIP), adults who have completed a primary series of childhood vaccinations (typically 5 doses of DTaP or DTP) should receive a single booster dose of Tdap if they have not previously received it. This is especially critical for healthcare personnel, such as a 55-year-old nurse, due to their increased risk of exposure to pertussis and the need to protect vulnerable patients (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.2 - Implement measures to prevent transmission of infectious agents). The Tdap vaccine, which protects against tetanus, diphtheria, and pertussis, is recommended once between ages 11-64, with a preference for administration in early adulthood (e.g., 19-26 years) or as soon as feasible for older adults, including this 55-year-old nurse, to ensure immunity against pertussis, which wanes over time. For individuals aged 65 and older, Tdap is still recommended if not previously received, though Tdap is preferred over Td (tetanus and diphtheria only) for healthcare workers to address pertussis risk.
Option B (two doses of Tdap vaccine at least 14 days apart) and Option C (two doses of Tdap vaccine at least 28 days apart) are not standard recommendations for adults with a complete childhood vaccination history. Multiple doses are typically reserved for individuals with incomplete primary series or specific high-risk conditions, not for this scenario. Option D (no additional vaccination is recommended) is incorrect because, even with a complete childhood series, a Tdap booster is advised for healthcare workers to maintain protection, especially given the nurse’s occupational exposure risks (CDC Immunization Schedules, 2024). After receiving the Tdap booster, a Td booster every 10 years is recommended to maintain tetanus and diphtheria immunity, but the initial Tdap dose is the priority for this nurse.
The recommendation for one Tdap dose aligns with CBIC’s emphasis on evidence-based immunization policies to prevent transmission of vaccine-preventable diseases in healthcare settings (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.1 - Collaborate with organizational leaders). This ensures the nurse is protected and contributes to herd immunity, reducing the risk of pertussis outbreaks in the healthcare environment.
Hand hygiene rates in the facility have been decreasing over time. The Infection Preventionist (IP) surveys staff and finds that hand dryness is the major reason for non-compliance. What step should the IP take?
Options:
Provide staff lotion in every patient room.
Provide a compatible lotion in a convenient location.
Allow staff to bring in lotion and carry it in their pockets.
Allow staff to bring in lotion for use at the nurses’ station and lounge.
Answer:
BExplanation:
Hand hygiene is a cornerstone of infection prevention, and declining compliance rates pose a significant risk for healthcare-associated infections (HAIs). The Certification Board of Infection Control and Epidemiology (CBIC) emphasizes improving hand hygiene adherence in the "Prevention and Control of Infectious Diseases" domain, aligning with the Centers for Disease Control and Prevention (CDC) "Guideline for Hand Hygiene in Healthcare Settings" (2002). The IP’s survey identifies hand dryness as the primary barrier, likely due to the frequent use of alcohol-based hand sanitizers or soap, which can dehydrate skin. The goal is to address this barrier effectively while maintaining infection control standards.
Option B, "Provide a compatible lotion in a convenient location," is the most appropriate step. The CDC and World Health Organization (WHO) recommend using moisturizers to mitigate skin irritation and dryness, which can improve hand hygiene compliance. However, the lotion must be compatible with alcohol-based hand rubs (e.g., free of petroleum-based products that can reduce sanitizer efficacy) and placed in accessible areas (e.g., near sinks or sanitizer dispensers) to encourage use without disrupting workflow. The WHO’s "Guidelines on Hand Hygiene in Health Care" (2009) suggest providing skin care products as part of a multimodal strategy to enhance adherence, making this a proactive, facility-supported solution that addresses the root cause.
Option A, "Provide staff lotion in every patient room," is a good intention but impractical and potentially risky. Placing lotion in patient rooms could lead to inconsistent use, contamination (e.g., from patient contact), or misuse (e.g., staff applying incompatible products), compromising infection control. The CDC advises against uncontrolled lotion distribution in patient care areas. Option C, "Allow staff to bring in lotion and carry it in their pockets," introduces variability in product quality and compatibility. Personal lotions may contain ingredients (e.g., oils) that inactivate alcohol-based sanitizers, and pocket storage increases the risk of contamination or cross-contamination, which the CDC cautions against. Option D, "Allow staff to bring in lotion for use at the nurses’ station and lounge," limits the intervention to non-patient care areas, reducing its impact on hand hygiene during patient interactions. It also shares the compatibility and contamination risks of Option C, making it less effective.
The CBIC Practice Analysis (2022) and CDC guidelines emphasize evidence-based interventions, such as providing approved skin care products in strategic locations to boost compliance. Option B balances accessibility, safety, and compatibility, making it the best step to address hand dryness and improve hand hygiene rates.
An infection preventionist is preparing a report about an outbreak of scabies in a long-term care facility. How would this information be displayed in an epidemic curve?
Options:
List case names, room numbers, and date the infestation was identified using a logarithmic scale.
List case medical record numbers and the number of days in the facility to date of onset, showing data in a scatter plot.
Prepare a bar graph with no patient identifiers showing the number of cases over a specific period of time.
Prepare a scatter plot by patient location showing case prevalence over a specific period of time.
Answer:
CExplanation:
An epidemic curve, commonly used in infection prevention and control to visualize the progression of an outbreak, is a graphical representation of the number of cases over time. According to the principles outlined by the Certification Board of Infection Control and Epidemiology (CBIC), an epidemic curve is most effectively displayed using a bar graph or histogram that tracks the number of new cases by date or time interval (e.g., daily, weekly) without revealing patient identifiers, ensuring compliance with privacy regulations such as HIPAA. Option C aligns with this standard practice, as it specifies preparing a bar graph with no patient identifiers, focusing solely on the number of cases over a specific period. This allows infection preventionists to identify patterns, such as the peak of the outbreak or potential sources of transmission, while maintaining confidentiality.
Option A is incorrect because listing case names and room numbers with a logarithmic scale violates patient privacy and is not a standard method for constructing an epidemic curve. Logarithmic scales are typically used for data with a wide range of values, but they are not the preferred format for epidemic curves, which prioritize clarity over time. Option B is also incorrect, as using medical record numbers and scatter plots to show days in the facility to onset does not align with the definition of an epidemic curve, which focuses on case counts over time rather than individual patient timelines or scatter plot formats. Option D is inappropriate because a scatter plot by patient location emphasizes spatial distribution rather than the temporal progression central to an epidemic curve. While location data can be useful in outbreak investigations, it is typically analyzed separately from the epidemic curve.
The CBIC emphasizes the importance of epidemic curves in the "Identification of Infectious Disease Processes" domain, where infection preventionists use such tools to monitor and control outbreaks (CBIC Practice Analysis, 2022). Specifically, the use of anonymized data in graphical formats is a best practice to protect patient information while providing actionable insights, as detailed in the CBIC Infection Prevention and Control (IPC) guidelines.
An infection preventionist has decided to perform surveillance for central line–associated bloodstream infections (CLABSIs) in the facility’s ICU. Which of the following is the MOST appropriate denominator to calculate risk-adjusted rates?
Options:
Total number of ICU patients
Total number of patients with central lines
Number of patients with infections who have central lines
Number of days patients have central lines in place
Answer:
DExplanation:
The Certification Study Guide (6th edition) emphasizes that device-associated infection rates must be calculated using time-at-risk denominators to accurately reflect patient exposure. For CLABSI surveillance, the most appropriate denominator is central line days, defined as the total number of days patients have one or more central lines in place during the surveillance period.
Using central line days accounts for both the presence and duration of exposure, which is critical for risk adjustment. The longer a central line remains in place, the greater the opportunity for microbial entry and bloodstream infection. This denominator allows for valid trend analysis over time and meaningful benchmarking with national surveillance systems that use standardized definitions and denominators.
The other options are incorrect because they fail to measure exposure accurately. Total ICU patients and total patients with central lines do not account for how long the device was present. Counting only patients who developed infections incorrectly places outcomes in the denominator, which invalidates rate calculations.
The study guide reinforces that numerators represent infection events, while denominators represent populations or time at risk. For CLABSI, the standard rate is expressed as infections per 1,000 central line days, a core concept frequently tested on the CIC exam.
Accurate denominator selection ensures valid surveillance, supports quality improvement efforts, and enables comparison with national benchmarks—making central line days the correct and most appropriate choice.
An infection preventionist wishes to compare central line infection rates with those of a national database. What consideration is of prime importance in making this comparison?
Options:
Comparable skin preparation prior to insertion
Use of identical types of central lines
Similar sizes of facilities in the database
Same definition of central line infection
Answer:
DExplanation:
The Certification Study Guide (6th edition) stresses that valid comparison of infection rates requires consistent surveillance definitions and methodologies. When comparing a facility’s central line–associated bloodstream infection (CLABSI) rates to those reported in a national database, the single most important consideration is the use of the same case definition for central line infection. Without standardized definitions, rate comparisons are unreliable and may lead to incorrect conclusions about performance.
National databases rely on precise, standardized criteria for what constitutes a CLABSI, including timing, clinical signs, laboratory confirmation, and attribution to a central line. If a facility applies different criteria—such as alternative timing windows, inclusion/exclusion rules, or diagnostic thresholds—the resulting rates may be artificially higher or lower than benchmark data. The study guide emphasizes that comparability hinges on alignment of numerators (cases) and denominators (central line days) using identical definitions.
The other options, while relevant to prevention practices or contextual understanding, are not primary requirements for valid comparison. Skin preparation methods and types of lines influence risk but do not ensure comparability of reported rates. Facility size can affect risk profiles, but standardized definitions allow for risk adjustment within databases.
This question reflects a core CIC exam principle: benchmarking is meaningful only when surveillance definitions are consistent. Ensuring alignment with national definitions is foundational to accurate performance evaluation and quality improvement.
Which of the following BEST demonstrates the effectiveness of a program targeted at reducing central-line associated bloodstream infections (CLABSIs) in an intensive care unit (ICU)?
Options:
A 25% decrease in the length of stay in the ICU related to CLABSIs
A 25% reduction in the incidence of CLABSIs over 6 months
A 30% decrease in total costs related to treatment of CLABSIs over 12 months
A 30% reduction in the use of antibiotic-impregnated central catheters over 6 months
Answer:
BExplanation:
Evaluating the effectiveness of a program to reduce central-line associated bloodstream infections (CLABSIs) in an intensive care unit (ICU) requires identifying the most direct and relevant measure of success. The Certification Board of Infection Control and Epidemiology (CBIC) emphasizes outcome-based assessment in the "Performance Improvement" and "Surveillance and Epidemiologic Investigation" domains, aligning with the Centers for Disease Control and Prevention (CDC) guidelines for infection prevention. The primary goal of a CLABSI reduction program is to decrease the occurrence of these infections, with secondary benefits including reduced length of stay, costs, and resource use.
Option B, "A 25% reduction in the incidence of CLABSIs over 6 months," is the best demonstration of effectiveness. The incidence of CLABSIs—defined by the CDC as the number of infections per 1,000 central line days—directly measures the program’s impact on the targeted outcome: preventing bloodstream infections associated with central lines. A 25% reduction over 6 months indicates a sustained decrease in infection rates, providing clear evidence that the intervention (e.g., improved insertion techniques, maintenance bundles, or staff education) is working. The CDC’s "Guidelines for the Prevention of Intravascular Catheter-Related Infections" (2017) and the National Healthcare Safety Network (NHSN) protocols prioritize infection rate reduction as the primary metric for assessing CLABSI prevention programs.
Option A, "A 25% decrease in the length of stay in the ICU related to CLABSIs," is a secondary benefit. Reducing CLABSI-related length of stay can improve patient outcomes and bed availability, but it is an indirect measure dependent on infection incidence. A decrease in length of stay could also reflect other factors (e.g., improved discharge planning), making it less specific to program effectiveness. Option C, "A 30% decrease in total costs related to treatment of CLABSIs over 12 months," reflects a financial outcome, which is valuable for justifying resource allocation. However, cost reduction is a downstream effect of decreased infections and may be influenced by variables like hospital pricing or treatment protocols, diluting its direct link to program success. Option D, "A 30% reduction in the use of antibiotic-impregnated central catheters over 6 months," indicates a change in practice but not necessarily effectiveness. Antibiotic-impregnated catheters are one prevention strategy, and reducing their use could suggest improved standard practices (e.g., chlorhexidine bathing), but it could also increase infection rates if not offset by other measures, making it an ambiguous indicator.
The CBIC Practice Analysis (2022) and CDC guidelines emphasize that the primary measure of a CLABSI prevention program’s success is a reduction in infection incidence, as it directly addresses patient safety and the program’s core objective. Option B provides the most robust and specific evidence of effectiveness over a defined timeframe.
What data should be collected and analyzed as part of a syndromic surveillance program?
Options:
Number of blood and urine cultures
Number of visits to physical therapy
Number of surgical procedures performed
Number of over-the-counter sales of flu remedies
Answer:
DExplanation:
The Certification Study Guide (6th edition) describes syndromic surveillance as a public health surveillance approach that focuses on the early detection of disease outbreaks by monitoring nonspecific indicators that precede formal diagnosis or laboratory confirmation. Rather than relying on confirmed cases, syndromic surveillance tracks patterns of symptoms, behaviors, or indirect data sources that may signal emerging health threats.
One key example emphasized in the study guide is the monitoring of over-the-counter (OTC) medication sales, such as flu and cold remedies. Increases in OTC sales can indicate a rise in respiratory illness within the community before patients seek medical care or receive laboratory testing. This early signal allows infection preventionists and public health officials to initiate investigations, preparedness measures, and targeted messaging sooner than traditional surveillance methods would allow.
The other options reflect data used in traditional or outcome-based surveillance, not syndromic surveillance. Blood and urine cultures require laboratory confirmation and occur later in the disease process. Physical therapy visits and surgical procedure counts are unrelated to early symptom detection and do not provide timely indicators of infectious disease trends.
CIC exam questions frequently test the distinction between traditional surveillance and syndromic surveillance. Recognizing that syndromic surveillance relies on early, indirect indicators of illness, such as OTC medication sales, is essential for accurate exam performance and effective outbreak preparedness.
The Sterile Processing Deportment alerts an infection preventionist that a load of surgical Instruments sterilized with high temperature steam:moist heat needs to be recalled. Which of the following Is the MOST likely reason for the recall?
Options:
Failure of the biological Indicator Bacillus subtilts
Failure of the biological Indicator Geobaciltus stearothermophilus
Placement of the biological Indicator on the bottom shelf over the d*an
Incorrect placement of the instruments In the tray
Answer:
BExplanation:
The most likely reason for the recall of a steam-sterilized load is the failure of the biological indicator (BI), specifically Geobacillus stearothermophilus, which is used to monitor high-temperature steam (moist heat) sterilization processes. This organism is the biological indicator of choice because it has high resistance to moist heat and thus serves as a reliable marker for sterilization efficacy.
The APIC Text and AAMI ST79 guidelines confirm that Geobacillus stearothermophilus is used for steam sterilization and that a failed BI indicates a failure in the sterilization process, which requires immediate action, including recalling all items sterilized since the last negative BI and reprocessing them. This is a crucial aspect of ensuring patient safety and preventing the use of potentially non-sterile surgical instruments.
According to the APIC Text:
"BIs are the only process indicators that directly monitor the lethality of a given sterilization process. [...] Geobacillus stearothermophilus spores are used to monitor steam sterilization..."
The CIC Study Guide (6th ed.) also specifies that:
"Evidence of sterilization failures (e.g., positive biological indicators) is the most common reason for a recall."
Additionally, it is noted:
“With steam sterilization, the instrument load does not need to be recalled for a single positive biological indicator test, with the exception of implantable objects.”
However, multiple positive BIs or BI failure confirmation does require a recall.
The incorrect options explained:
A. Bacillus subtilis – This is not used in steam sterilization but rather in dry heat or EO processes.
C. Placement of the biological indicator on the bottom shelf over the drain – While incorrect placement can lead to test failure, the recall is prompted by BI failure, not just placement.
D. Incorrect placement of instruments – This can cause sterilization failure but is not the direct trigger for a recall unless it leads to a failed BI.
A healthcare worker experiences a percutaneous exposure to a patient with untreated HIV. The next step is to:
Options:
Initiate HIV post-exposure prophylaxis (PEP) within 2 hours.
Wait for HIV test results before starting treatment.
Offer post-exposure prophylaxis only if symptoms develop.
Retest for HIV after 6 months before deciding on PEP.
Answer:
AExplanation:
HIV post-exposure prophylaxis (PEP) should be initiated within 2 hours to be most effective.
Waiting for results (B) delays critical treatment.
PEP should always be offered after high-risk exposure, not only if symptoms develop (C).
Retesting after 6 months (D) is recommended but should not delay PEP initiation.
CBIC Infection Control References:
APIC Text, "Bloodborne Pathogens and PEP," Chapter 11.
Healthcare-associated transmission of methicillin-resistant Staphylococcus aureus (MRSA) is commonly associated with which of the following?
Options:
Improper ventilation
MRSA-infected patients
Inadequate hand hygiene
MRSA-colonized healthcare workers
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies inadequate hand hygiene as the most common and significant factor associated with healthcare-associated transmission of methicillin-resistant Staphylococcus aureus (MRSA). MRSA is primarily transmitted via direct contact, most often through the hands of healthcare personnel after contact with colonized or infected patients or contaminated environmental surfaces.
While MRSA-infected or colonized patients serve as reservoirs for the organism, transmission does not occur unless there is a breakdown in infection prevention practices, particularly hand hygiene. Numerous studies and surveillance findings cited in the Study Guide demonstrate that adherence to hand hygiene protocols—before and after patient contact, after contact with bodily fluids, and after contact with the patient environment—is the single most effective measure to reduce MRSA spread within healthcare facilities.
Improper ventilation (Option A) is associated with airborne pathogens, not MRSA, which is not transmitted via the airborne route. MRSA-colonized healthcare workers (Option D) are far less commonly implicated in transmission than transient hand contamination, and routine screening of staff is not recommended except during specific outbreak investigations. Option B describes a reservoir, not the primary mechanism of transmission.
For CIC® exam purposes, this question reinforces a foundational principle of infection prevention: failure to perform appropriate hand hygiene is the leading cause of healthcare-associated MRSA transmission, making it the correct and best answer.
The infection preventionist recognizes that construction barriers are a key component of the Infection Control Risk Assessment (ICRA). The MOST important factor to consider is that construction barriers should:
Options:
Be constructed to withstand normal heating, ventilation, and air conditioning (HVAC) airflow rates.
Provide sealed covers for air intakes and exhausts.
Be able to contain dust or infectious microorganisms generated by the project.
Have walk-off mats that are changed daily.
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that the primary purpose of construction barriers within an Infection Control Risk Assessment (ICRA) is to prevent the dissemination of dust and potentially infectious microorganisms generated during construction, renovation, or maintenance activities. Construction activities can aerosolize fungal spores (such as Aspergillus), bacteria, and other particulate matter that pose a significant risk to immunocompromised patients and other vulnerable populations.
Barriers must therefore be designed and maintained to effectively contain dust and microorganisms at the source, preventing their migration into occupied patient care areas. This containment function is the cornerstone of infection prevention during construction and directly aligns with ICRA goals of risk reduction and patient safety.
While the other options describe supportive or secondary considerations, they are not the most critical factor. Withstanding HVAC airflow (Option A) is important, but it serves the larger goal of containment. Sealing air intakes and exhausts (Option B) is a specific engineering control that may be used as part of containment strategies but does not define the primary purpose of barriers. Walk-off mats (Option D) are useful adjunctive controls but are insufficient alone to prevent airborne transmission of contaminants.
For CIC® exam preparation, it is essential to recognize that containment of dust and infectious agents is the defining function of construction barriers within an ICRA, and all other measures support this central objective.
==========
An infection preventionist is asked to recommend a product for disinfection of bronchoscopes. Which of the following agents would be appropriate?
Options:
Iodophor
Alcohol
Phenolic
Peracetic acid
Answer:
DExplanation:
The correct answer is D, "Peracetic acid," as this agent is appropriate for the disinfection of bronchoscopes. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, bronchoscopes are semi-critical devices that require high-level disinfection (HLD) to eliminate all microorganisms except high levels of bacterial spores, as they come into contact with mucous membranes but not sterile tissues. Peracetic acid is recognized by the Centers for Disease Control and Prevention (CDC) and the Association for the Advancement of Medical Instrumentation (AAMI) as an effective high-level disinfectant for endoscopes, including bronchoscopes, due to its broad-spectrum antimicrobial activity, rapid action, and compatibility with the delicate materials (e.g., optics and channels) of these devices (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.3 - Ensure safe reprocessing of medical equipment). It is commonly used in automated endoscope reprocessors, ensuring thorough disinfection when combined with proper cleaning and rinsing protocols.
Option A (iodophor) is typically used for intermediate-level disinfection and skin antisepsis, but it is not sufficient for high-level disinfection of bronchoscopes unless specifically formulated and validated for this purpose, which is uncommon. Option B (alcohol) is effective against some pathogens but evaporates quickly, fails to penetrate organic material, and is not recommended for HLD of endoscopes due to potential damage to internal components and inadequate sporicidal activity. Option C (phenolic) is suitable for surface disinfection but lacks the efficacy required for high-level disinfection of semi-critical devices like bronchoscopes, as it does not reliably eliminate all microbial threats, including mycobacteria.
The selection of peracetic acid aligns with CBIC’s emphasis on evidence-based reprocessing practices to prevent healthcare-associated infections (HAIs) associated with endoscope use (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.4 - Implement environmental cleaning and disinfection protocols). This choice ensures patient safety by adhering to manufacturer and regulatory guidelines, such as those in AAMI ST91 (AAMI ST91:2015, Flexible and semi-rigid endoscope processing in health care facilities).
Which of the following processes would the catheter-associated urinary tract infection (CAUTI) improvement team choose as a process indicator to reduce CAUTI?
Options:
Documentation of indication for catheter placement
Rate of catheter-associated urinary tract infections
Reduction of catheter insertions per month
Rate of asymptomatic bacteriuria
Answer:
AExplanation:
A process indicator measures whether staff are reliably performing evidence-based practices that prevent infection (i.e., how well we do what we intend to do). For CAUTI prevention, a core, guideline-supported strategy is to use indwelling urinary catheters only for appropriate indications and remove them as soon as they are no longer needed. Because inappropriate placement is a major driver of unnecessary catheter days (and therefore CAUTI risk), tracking whether the clinical indication is documented at insertion is a practical, auditable process measure that directly reflects adherence to appropriate-use policies. The CDC CAUTI prevention toolkit lists “compliance with documentation of catheter …” as an example of a process measure, aligning with performance measurement approaches recommended for CAUTI prevention.
In contrast, the CAUTI rate (option B) is an outcome measure, not a process measure. “Reduction of catheter insertions per month” (option C) reflects volume/usage trends rather than direct compliance with a specific practice, and “rate of asymptomatic bacteriuria” (option D) is not a recommended target metric for CAUTI prevention and routine screening for ASB is discouraged in CAUTI guidance.
An infection preventionist is writing a policy about prevention of intravascular device infection. Which of the following is important for healthcare personnel to know as part of central line insertion and maintenance procedures?
Options:
Change the central line every seven days.
Use maximum sterile barrier precautions for the line insertion.
The femoral site is the preferred site of insertion in an adult patient.
Use 70% isopropyl alcohol for skin preparation before line insertion.
Answer:
BExplanation:
The Certification Study Guide (6th edition) identifies the use of maximum sterile barrier (MSB) precautions during central line insertion as a cornerstone practice for preventing intravascular device–associated infections, including central line–associated bloodstream infections (CLABSIs). MSB precautions include wearing a cap, mask, sterile gown, and sterile gloves, and using a large sterile drape to fully cover the patient during line insertion. These measures significantly reduce the risk of introducing skin flora and environmental microorganisms into the bloodstream at the time of catheter placement.
The study guide emphasizes that the highest risk for contamination occurs during insertion, making strict aseptic technique essential. MSB precautions are a required element of evidence-based central line insertion bundles and are consistently associated with reduced CLABSI rates when reliably implemented.
The other options reflect outdated or incorrect practices. Routine scheduled replacement of central lines every seven days is not recommended and does not reduce infection risk. The femoral vein is not the preferred insertion site in adults due to higher infection risk compared to subclavian or internal jugular sites. While alcohol is used during hub disinfection, chlorhexidine-based antisepsis (preferably chlorhexidine with alcohol) is recommended for skin preparation—not alcohol alone.
This question highlights a core CIC exam concept: standardized insertion practices using maximum sterile barriers are among the most effective strategies for preventing intravascular device infections.
Which of the following is the BEST strategy for reducing bloodstream infections associated with central venous catheters?
Options:
Routine replacement of central lines every 7 days.
Use of chlorhexidine-impregnated dressings.
Daily blood cultures for patients with central lines.
Use of povidone-iodine instead of chlorhexidine for skin antisepsis.
Answer:
BExplanation:
Chlorhexidine-impregnated dressings reduce central line-associated bloodstream infections (CLABSI) by preventing bacterial colonization.
Routine catheter replacement (A) increases insertion risks without reducing infections.
Daily blood cultures (C) are unnecessary and lead to false positives.
Povidone-iodine (D) is less effective than chlorhexidine for skin antisepsis.
CBIC Infection Control References:
APIC Text, "CLABSI Prevention Measures," Chapter 10.
An infection preventionist has been asked to participate in a process improvement team to standardize disinfection and sterilization practices. Team activities should include all of the following EXCEPT:
Options:
Observing disinfection and sterilization practices.
Asking central supply and operating room managers to join the team.
Performing a literature review on central supply and sterilization.
Conducting outcome measurement after all changes are implemented.
Answer:
DExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that effective process improvement relies on a structured, data-driven approach that includes baseline assessment, intervention, and ongoing evaluation. A key principle of quality improvement is that outcomes must be measured before and after changes are implemented in order to determine whether an intervention resulted in improvement.
Option D is the correct “EXCEPT” choice because limiting outcome measurement to only after changes are implemented prevents meaningful comparison and makes it impossible to determine effectiveness. Without baseline data, improvements cannot be quantified, trends cannot be assessed, and unintended consequences may go unrecognized. The Study Guide stresses that baseline measurements are essential to evaluate process performance and to support evidence-based decision-making.
Options A, B, and C are all appropriate and expected activities. Direct observation helps identify workflow gaps and variation in practice. Inclusion of central supply and operating room leadership ensures multidisciplinary engagement and operational insight. Conducting a literature review supports alignment with current evidence, standards, and best practices for disinfection and sterilization.
For the CIC® exam, it is important to recognize that continuous measurement throughout the improvement cycle—not only after implementation—is required for successful standardization and sustainability of infection prevention practices.
What is a characteristic of immediate-use steam sterilization?
Options:
Alternative to purchasing expensive instrument sets.
Can be used for the following surgery if properly stored.
Substitute for maintaining sufficient amounts of sterile instruments.
Performed in emergencies where cleaning is the most critical step.
Answer:
CExplanation:
The correct answer is C, "Substitute for maintaining sufficient amounts of sterile instruments," as this is a characteristic of immediate-use steam sterilization (IUSS). According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, IUSS, formerly known as flash sterilization, is a process designed to rapidly sterilize items that are needed urgently when pre-sterilized inventory is unavailable or insufficient. It serves as a temporary solution to address gaps in sterile instrument availability, such as during unexpected surges in surgical demand or equipment shortages, provided strict protocols are followed (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.3 - Ensure safe reprocessing of medical equipment). However, IUSS is not a routine practice and should be minimized due to its limitations, including the lack of immediate biologic indicator results.
Option A (alternative to purchasing expensive instrument sets) is incorrect because IUSS is not intended as a cost-saving measure or a replacement for acquiring necessary equipment; it is a contingency process. Option B (can be used for the following surgery if properly stored) is misleading, as IUSS items are intended for immediate use and not for storage or use in subsequent procedures, which requires standard sterilization cycles with proper packaging and validation. Option D (performed in emergencies where cleaning is the most critical step) overemphasizes cleaning and mischaracterizes IUSS; while cleaning is a critical initial step, the process is defined by its rapid sterilization for emergency use, not solely by cleaning priority.
The characteristic of substituting for insufficient sterile instruments aligns with CBIC’s focus on ensuring safe reprocessing practices while acknowledging the practical challenges in healthcare settings (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.5 - Evaluate the environment for infection risks). This is supported by AAMI ST79, which outlines IUSS as a last-resort measure to maintain surgical readiness (AAMI ST79:2017).
When describing the differences between colonization and infection, the infection preventionist should define colonization as
Options:
Tissue invasion by organisms without tissue damage.
Tissue injury by normal flora associated with local reaction.
The presence and growth of organisms without tissue invasion.
The presence and growth of opportunistic organisms with cellular change.
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) clearly distinguishes colonization from infection, a foundational concept in infection prevention and healthcare epidemiology. Colonization is defined as the presence and multiplication of microorganisms on or within a host without tissue invasion, damage, or clinical signs of disease. Individuals who are colonized do not exhibit symptoms and typically do not mount an inflammatory response.
Option C accurately reflects this definition and is the correct answer. Colonized organisms may be part of normal flora or may be potentially pathogenic organisms such as Staphylococcus aureus or multidrug-resistant organisms. Although colonization does not cause illness, colonized individuals can serve as reservoirs for transmission and may later develop infection if host defenses are compromised.
Option A is incorrect because tissue invasion, even without visible damage, represents infection rather than colonization. Option B describes infection caused by normal flora with an inflammatory response. Option D includes cellular change, which indicates tissue response and therefore infection.
For the CIC® exam, it is essential to understand that colonization involves microbial presence without host response, while infection requires tissue invasion and a corresponding inflammatory or immune reaction. This distinction is critical for surveillance definitions, isolation decisions, antimicrobial stewardship, and patient education.
Which of the following anti-infective materials is used on endotracheal tubes, urine catheters, and intravascular catheters?
Options:
Silver
Copper
Chromium
Zinc
Answer:
AExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies silver as an anti-infective material commonly incorporated into medical devices such as endotracheal tubes, urinary catheters, and intravascular catheters. Silver has broad-spectrum antimicrobial properties against bacteria, fungi, and some viruses. When used as a coating or impregnated material, silver ions disrupt microbial cell membranes, interfere with enzyme systems, and inhibit replication—thereby reducing microbial colonization and biofilm formation on device surfaces.
Device-associated infections often originate from colonization of indwelling devices. Silver-coated or silver-impregnated devices are intended to reduce the risk of healthcare-associated infections by limiting early microbial adherence and growth, particularly during the highest-risk period shortly after device insertion. Examples include silver alloy urinary catheters for CAUTI prevention and silver-coated endotracheal tubes designed to reduce ventilator-associated events.
The other options listed are not used in this context. Copper has antimicrobial properties but is not commonly used in indwelling medical devices. Chromium is used for corrosion resistance in alloys, not for infection prevention. Zinc plays roles in wound care and topical formulations but is not standard for catheter or tube coatings.
For CIC® exam preparation, recognizing silver as the anti-infective material used in multiple indwelling devices is important, as it reflects evidence-based strategies aimed at reducing device-associated infection risk.
==========
The infection preventionist (IP) is working with the Product Evaluation Committee to select a sporicidal disinfectant for Clostridioides difficile. An effective disinfectant for the IP to recommend is
Options:
quaternary ammonium compound.
phenolic.
isopropyl alcohol.
sodium hypochlorite.
Answer:
DExplanation:
The correct answer is D, "sodium hypochlorite," as it is an effective sporicidal disinfectant for Clostridioides difficile that the infection preventionist (IP) should recommend. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, Clostridioides difficile (C. difficile) is a spore-forming bacterium responsible for significant healthcare-associated infections (HAIs), and its spores are highly resistant to many common disinfectants. Sodium hypochlorite (bleach) is recognized by the Centers for Disease Control and Prevention (CDC) and the Environmental Protection Agency (EPA) as a sporicidal agent capable of inactivating C. difficile spores when used at appropriate concentrations (e.g., 1:10 dilution of household bleach) and with the recommended contact time (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.4 - Implement environmental cleaning and disinfection protocols). This makes it a preferred choice for environmental disinfection in outbreak settings or areas with known C. difficile contamination.
Option A (quaternary ammonium compound) is effective against many bacteria and viruses but lacks sufficient sporicidal activity against C. difficile spores, rendering it inadequate for this purpose. Option B (phenolic) has broad-spectrum antimicrobial properties but is not reliably sporicidal and is less effective against C. difficile spores compared to sodium hypochlorite. Option C (isopropyl alcohol) is useful for disinfecting surfaces and killing some pathogens, but it is not sporicidal and evaporates quickly, making it ineffective against C. difficile spores.
The IP’s recommendation of sodium hypochlorite aligns with CBIC’s emphasis on selecting disinfectants based on their efficacy against specific pathogens and adherence to evidence-based guidelines (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.5 - Evaluate the environment for infection risks). Proper use, including correct dilution and contact time, is critical to ensure effectiveness, and the IP should collaborate with the Product Evaluation Committee to ensure implementation aligns with safety and regulatory standards (CDC Guidelines for Environmental Infection Control in Healthcare Facilities, 2019).
An infection preventionist (IP) is tasked with identifying if the Intensive Care Unit’s (ICU) central line–associated bloodstream infection (CLABSI) prevention practices are consistent with current best practices. Which of the following quality improvement tools should the IP construct?
Options:
Gap analysis
Root cause analysis
Failure mode and effect analysis (FMEA)
Strengths, weaknesses, opportunities, and threats (SWOT) analysis
Answer:
AExplanation:
The Certification Study Guide (6th edition) clearly distinguishes among quality improvement tools based on their purpose and timing. When the goal is to determine whether current practices align with evidence-based standards or best practices, the most appropriate tool is a gap analysis. A gap analysis systematically compares current state practices—such as ICU CLABSI prevention policies, procedures, and compliance data—with the desired state, which is defined by nationally recognized guidelines and best practices.
The study guide emphasizes that gap analysis is particularly useful for program evaluation, policy review, and baseline assessment before implementing improvements. In this scenario, the IP is not responding to an adverse event, nor is the IP proactively predicting failures, but rather assessing alignment with best practices, which is the core function of a gap analysis.
The other tools serve different purposes. Root cause analysis (RCA) is used after an adverse event (such as a CLABSI) to identify contributing factors. Failure mode and effect analysis (FMEA) is a prospective risk assessment tool used to anticipate where processes might fail. SWOT analysis is a strategic planning tool and is not sufficiently specific for evaluating compliance with infection prevention standards.
Because CIC exam questions frequently test the ability to select the right tool for the right situation, recognizing gap analysis as the appropriate choice in this context is essential.
An infection control manager is training a new infection preventionist. In discussing surveillance strategies, which of the following types of hospital infection surveillance usually provides maximum benefit with minimum resources?
Options:
High-risk patient focus
Antibiotic monitoring
Prevalence surveys
Nursing care plan review
Answer:
AExplanation:
A high-risk patient focus maximizes benefits while minimizing resource use in infection surveillance.
Step-by-Step Justification:
Efficiency of High-Risk Surveillance:
Targeting ICU, immunocompromised patients, or surgical units helps detect infections where the risk is highest, leading to earlier interventions.
Resource Allocation:
Full hospital-wide surveillance is resource-intensive; focusing on high-risk groups is more efficient.
Why Other Options Are Incorrect:
B. Antibiotic monitoring:
Important for stewardship, but not the primary focus of infection surveillance.
C. Prevalence surveys:
Snapshot data only; does not provide ongoing monitoring.
D. Nursing care plan review:
Less direct in identifying infection trends.
CBIC Infection Control References:
APIC Text, "Surveillance Strategies for Infection Prevention".
An infection preventionist (IP) is reviewing blood cultures and notices several results with Arcanobacterium, coagulase-negative Staphylococcus, and Corynebacterium. What action is needed from the IP?
Options:
Disregard the results.
Call the Medical Staff Officer and declare there is an outbreak.
Work up each case as a healthcare-acquired bloodstream infection.
Collaborate with the lab manager to determine if there are trends or changes in practice.
Answer:
DExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that certain organisms commonly recovered from blood cultures—such as Arcanobacterium, coagulase-negative Staphylococcus, and Corynebacterium—are frequently associated with skin contamination rather than true bloodstream infection. When multiple blood cultures yield these organisms, the infection preventionist must assess whether the findings represent contamination related to collection practices rather than immediately assuming infection or outbreak.
The most appropriate action is to collaborate with the laboratory manager and clinical teams to evaluate potential trends, specimen collection techniques, and changes in practice. This includes reviewing blood culture contamination rates, assessing skin antisepsis procedures, evaluating staff competency, and determining whether there has been an increase associated with a specific unit, shift, or collection method. Surveillance data and laboratory quality indicators are essential tools in this evaluation.
Option A is incorrect because results should never be disregarded without assessment. Option B is premature, as the organisms listed are not typical outbreak pathogens and require further analysis before escalation. Option C is inappropriate because these organisms do not automatically meet criteria for healthcare-associated bloodstream infection without supporting clinical evidence.
This scenario reflects a core CIC® exam concept: infection preventionists must apply epidemiologic principles, collaborate with laboratory services, and use data-driven analysis to differentiate contamination from infection and to guide quality improvement efforts.
=========
Which of the following represents the most effective strategy for preventing Clostridioides difficile transmission in a healthcare facility?
Options:
Daily environmental cleaning with quaternary ammonium compounds.
Strict antimicrobial stewardship to limit unnecessary antibiotic use.
Universal C. difficile screening on admission for high-risk patients.
Routine use of alcohol-based hand rub for hand hygiene after patient contact.
Answer:
BExplanation:
Antimicrobial stewardship is the most effective strategy to reduce C. difficile infections (CDI) by limiting the use of broad-spectrum antibiotics.
Quaternary ammonium disinfectants (A) are ineffective against C. difficile spores; bleach-based disinfectants are preferred.
Routine screening (C) is not cost-effective for prevention.
Alcohol-based hand rubs (D) do not kill C. difficile spores; soap and water should be used.
CBIC Infection Control References:
APIC Text, "C. difficile Prevention Strategies," Chapter 9.

